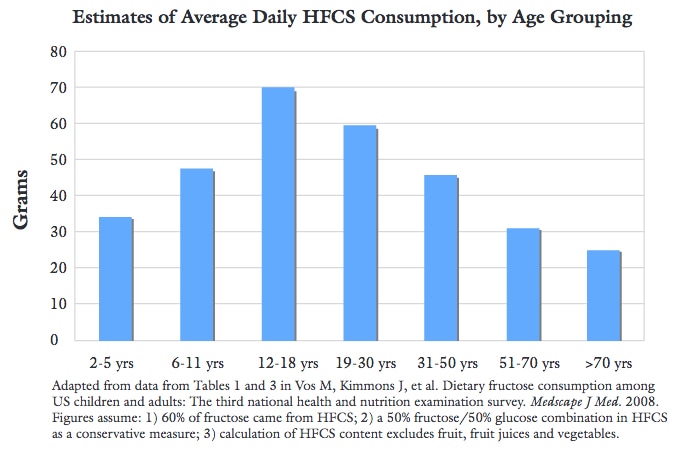
A group of researchers evaluated HFCS from three different manufacturers and determined that the 9 of the 20 samples were found to contain levels of mercury ranging from below a detection limit of 0.005 to 0.570 micrograms mercury per gram of HFCS (Dufault et al., 2009). A seperate group of researchers detected mercury in about one-third (or 17) of 55 popular brand-name food and beverage products where HFCS is the first or second highest labeled ingredient - including products by Quaker, Hershey's, Kraft, Coca-Cola and many more (Wallinga, Sorensen, Mottl & Yablon, 2009). Many of these products are marketed towards children, a huge portion of population that is more vulnerable to the toxic effects of mercury. How Did Mercury Get Into HFCS? tyHFCS has been made and is continued to be made using “mercury-grade” caustic soda. Caustic soda is produced by a mercury cell process which is contaminated with as much as 1 ppm, in some cases. Mercury contamination within soft drinks or drink mixes made from this caustic soda has been acknowledged by the National Association of Clean Water Agencies as early as 2000. Other common food ingredients derived from this mercury cell chlor-alkali plants process include citric acid and sodium benzoate, a food preservative found in many foods also containing HFCS. These other ingredients have not yet been tested for mercury contamination. Other common household products made from caustic soda also may be contaminated with low ppb levels of mercury, including shampoo, toilet tissue, bleach and toothpaste (Wallinga, Sorensen, Mottl & Yablon, 2009). What Is Caustic Soda? Chlorine is a widespread chemical building block used to make a vairety of products. Since 1884, one process for synthesizing chlorine has been to pump saltwater through a tank of mercury, also known as a mercury “cell.” These mercury cell (chlor-alkali) processing plants average 56 mercury cells each, with as much as 8,000 pounds of mercury per cell. Today, the chlorine industry remains the largest intentional consumer of mercury. While the mercury in the plants is reportedly left behind and reused, this process does not account for how volatile mercury is, and it is well known that contamination occurs throughout the process. These processing plants do not only produce chlorine, but a number of other products as well, including caustic soda (lye), sodium hypochlorite (bleach) and hydrochloric acid. Regardless of the product created, it and the wastewater end up being contaminated with mercury. Newer technology does exist for producing chlorine without mercury. However, caustic soda that is destined for the production of HFCS comes from either mercury cell or other processing plants (Wallinga, Sorensen, Mottl & Yablon, 2009). How Is HFCS Made? HFCS is produced in a highly specialized, industrial process using a myriad of enzymes and other materials, however either membrane-grade or mercury-grade caustic soda can be used. When creating HFCS, caustic soda helps separate the corn starch from the corn kernel. Caustic soda and hydrochloric acid can be used throughout the process to maintain a pH balance. Ultimately, the mercury-contaminated caustic soda may contaminate whatever products that are made from it, like HFCS. Indirectly, caustic soda also can contaminate the final food products to which HFCS is added (Wallinga, Sorensen, Mottl & Yablon, 2009). Avoid HFCS At All Costs Hidden in a wide variety of food-like products, including soda and ketchup, HFCS is prevalent throughout developed nations. Considering that American's consume roughly 50 grams of HFCS each day (equivalent to 12.5 teaspoons of sugar), even if a small portion of that is contaminated with mercury, the act should be concerning. Not to mention that HFCS, let alone added sugar, is linked a myriad of health deficits, including but not limited to increased risk of obesity, metabolic syndrome, cardiovascular disease, nonalcholic fatty liver disease, and other serious health conditions (Rippe & Etherton, 2012). There are enough reasons as it is to avoid HFCS, even if it hasn't been observed to be contaminated with mercury. References Dufault, R., LeBlanc, B., Schnoll, R., Cornett, C., Schweitzer, L., & Wallinga, D. et al. (2009). Mercury from chlor-alkali plants: measured concentrations in food product sugar. Environmental Health, 8(1). http://dx.doi.org/10.1186/1476-069x-8-2
Kaur, S., Kamli, M. R., & Ali, A. (2011). Role of arsenic and its resistance in nature. Canadian Journal Of Microbiology, 57(10), 769-774. doi:10.1139/w11-062 Rippe, J., & Etherton, P. (2012). Fructose, Sucrose, and High Fructose Corn Syrup: Modern Scientific Findings and Health Implications. Advances In Nutrition: An International Review Journal, 3(5), 739-740. http://dx.doi.org/10.3945/an.112.002600 Wallinga, D., Sorensen, J., Mottl, P., & Yablon, B. (2009). Not so sweet: Missing mercury and high fructose corn syrup: Mnstitute for agriculture and trade policy. Alternative Medicine Review, 14(2), 110.
0 Comments
Leave a Reply. |
The Awareness domain contains research, news, information, observations, and ideas at the level of self in an effort to intellectualize health concepts.
The Lifestyle domain builds off intellectual concepts and offers practical applications.
Taking care of yourself is at the core of the other domains because the others depend on your health and wellness.
Archives
April 2024
Categories
All
|

 RSS Feed
RSS Feed

