|
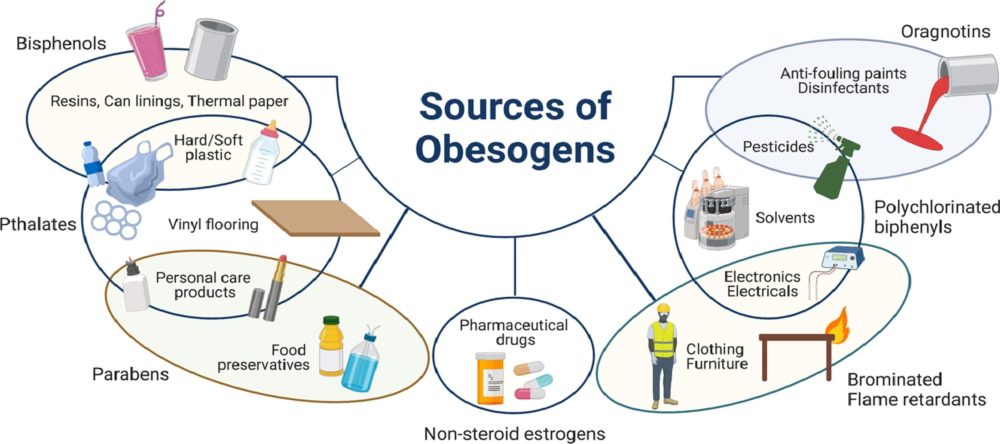
In our modern world, where convenience often comes at a cost, the prevalence of obesogens – chemicals that disrupt the body's normal metabolism and contribute to weight gain – has emerged as a growing concern. From everyday products to industrial pollutants, obesogens permeate our environment, exerting subtle yet profound effects on our health and well-being. Commonly encountered obesogensAmong the many obesogens encountered in daily life, several stand out for their widespread use and potential health impacts:
Mechanisms of ActionObesogens exert their effects through various mechanisms, including:
Disruption of Metabolism via MitochondriaObesogens, through their pervasive presence in our environment, exert insidious effects on metabolic function, including the intricate workings of mitochondria – the cellular powerhouses responsible for energy production. By disrupting mitochondrial function, obesogens can contribute to metabolic dysregulation and, ultimately, weight gain. Mitochondria play a central role in energy metabolism, converting nutrients into adenosine triphosphate (ATP), the primary source of cellular energy. However, exposure to obesogens can impair mitochondrial function through various mechanisms, including:
The disruption of mitochondrial function by obesogens can have profound implications for metabolic health and contribute to obesity through several pathways:
causative relationship with health conditionsThe impact of obesogens on human health extends beyond weight gain, with associations documented with various health conditions, including:
Additionally, obesogens are highly related to the following health conditions and physiologic imbalances:
Unraveling the Role of Dysfunctional Adipose TissueRelatively little is known about the extent to which obesogen exposure programs dysfunctional adipose tissue that may store but not mobilize fat. However, emerging evidence suggests that obesogens may contribute to adipocyte dysfunction, leading to altered fat storage and metabolism. One potential underlying factor is suboptimal liver detoxification pathways due to inadequate micronutrient cofactors. Inadequate levels of essential micronutrients, such as vitamins and minerals, can impair liver detoxification pathways responsible for metabolizing and eliminating obesogens from the body. As a result, obesogens may accumulate in adipose tissue, disrupting metabolic function and contributing to weight gain. Additionally, micronutrient deficiencies can compromise mitochondrial function, further exacerbating metabolic dysfunction and obesity risk. A Layman's Overview of Obesogens: Redefining the Weight Loss ParadigmIn the quest for weight loss, many of us often find ourselves fixating on calorie counting, fad diets, or intense workout regimens. However, what if I told you that the key to achieving a healthy weight isn't solely about shedding pounds but rather fixing your metabolism? Enter obesogens – a lesser-known yet influential factor in the obesity epidemic. As mentioned, obesogens are chemicals found in our environment, ranging from pesticides and plastics to food additives and personal care products. These substances have the uncanny ability to disrupt our body's natural weight-regulating mechanisms, leading to weight gain and metabolic dysfunction. Instead of solely blaming calories in versus calories out, it's essential to recognize the role obesogens play in shaping our metabolism. The Better Question: Fixing MetabolismRather than constantly asking ourselves, "How do I lose weight?" a more pertinent question would be: "How do I fix my metabolism?" Fixing metabolism involves addressing the root cause of weight gain – obesogen exposure and metabolic disruption. By eliminating or reducing our exposure to obesogens and ensuring our bodies receive essential micronutrients, we can optimize metabolic function and promote overall health. The Two-Fold SolutionTo achieve optimal health and maintain a healthy weight, a two-fold approach is necessary: 1. Reduce Toxin Exposure: Minimize exposure to obesogens by making conscious choices in our daily lives. This includes opting for organic produce, using natural cleaning and personal care products, and avoiding plastic containers and food packaging whenever possible. By participating in a structured evidenced-based detoxification program, we in turn lower our toxic burden, and we can mitigate the adverse effects of obesogens on our metabolism. 2. Consume Micronutrients: Vital micronutrients, such as vitamins and minerals, serve as essential cofactors in metabolic pathways. Ensuring adequate intake of these micronutrients through a balanced diet rich in fruits, vegetables, whole grains, and lean proteins can support optimal metabolic function. Additionally, supplementation may be necessary to address any deficiencies and promote metabolic health. The conventional approach to weight loss often overlooks the critical role obesogens play in metabolic dysfunction. Instead of solely focusing on calorie restriction or intense exercise, shifting our focus to fixing metabolism through toxin reduction and micronutrient consumption offers a more holistic and sustainable solution to achieving optimal health. By addressing the underlying factors contributing to metabolic disruption, we can pave the way for lasting weight management and overall well-being. the harm of environmental toxinsThe disruption of metabolic and mitochondrial function by obesogens represents a significant public health concern, with implications for obesity and metabolic disease. By understanding the mechanisms through which obesogens impair mitochondrial function and contribute to weight gain, researchers can develop targeted interventions to mitigate their adverse effects on metabolic health. Moreover, addressing underlying factors such as suboptimal liver detoxification pathways and micronutrient deficiencies is essential in combating the detrimental impact of obesogens on metabolic function and obesity prevalence. The pervasive presence of obesogens in our environment underscores the need for greater awareness and regulation of these harmful chemicals. By minimizing exposure to obesogens and advocating for safer alternatives, we can mitigate their adverse effects on human health and combat the rising tide of obesity and metabolic disease. As we navigate the complexities of modern living, vigilance and informed consumer choices are essential in safeguarding our health and well-being against the hidden threats of obesogens. Taking Action: The Integral Wellness ProgramFor those seeking tangible solutions to combat the effects of obesogens and improve their overall well-being, the Integral Wellness Program offers a comprehensive approach to optimizing health and vitality. This flagship service provides personalized guidance and support in key areas of movement, nutrition, and lifestyle to directly enhance quality of life. Online/In-Person Guidance One of the standout features of the Integral Wellness Program is its flexibility, offering both online and in-person consultations tailored to individual preferences and needs. Whether you prefer the convenience of virtual sessions or the hands-on approach of in-person coaching, our team of experienced wellness professionals is dedicated to supporting you every step of the way. Movement, Nutrition, and Lifestyle The Integral Wellness Program takes a holistic approach to health, addressing modifiable factors and behaviors in three core areas:
Augmenting the Health Process By participating in the Integral Wellness Program, you'll not only gain valuable knowledge and skills to navigate the challenges of modern living but also receive ongoing support and accountability to stay on track towards your health goals. Through targeted interventions aimed at eliminating obesogen exposure and promoting healthy behaviors, you can unlock your body's full potential and thrive in all aspects of life. The Integral Wellness Program offers a transformative journey towards optimal health and vitality. By prioritizing movement, nutrition, and lifestyle modifications, participants can take proactive steps to combat the effects of obesogens and reclaim control over their well-being. With the guidance and support of our dedicated wellness professionals, you'll embark on a path of self-discovery, empowerment, and lasting transformation. references
0 Comments
The environment that we live in is toxic. It is worrisome to think that the status quo has occurred with the help of corporations knowingly dumping harmful chemicals into the environment.
The Environmental Working Group (EWG) has studied the current state of our world in great detail and has discovered that before a child is even born they already have approximately 287 toxins in their blood and tissues. These results came from 10 newborns whose parents gave permission to have their toxins measured at birth. The results of this study indicate that an average of 200 chemicals was found in each newborn. Of the toxins tested, 47 were consumer ingredients such as cosmetics, 212 were industrial and pesticide byproducts. In this study, only around 400 total chemicals were actually tested for - thousands of others may have been found if larger parameters were used. Many of the toxins measured in the newborns included plastics, flame retardants, and other chemicals that disrupt brain function, IQ, hormones, and the nervous system of the child. Some of the toxins observed like DDT, have actually been banned since 1972 (over 3 decades ago), but are still being measured in laboratory samples. Certain chemicals never fully degrade in the environment. So, there is not question about it, the environment we live in is toxic. All of us have disease-creating toxins inside of our bodies, the question boils down to which ones and how much. But there are some solutions: lab tests and detoxification.
More than 2000 references on the biological responses to radio frequency (RF) and microwave radiation, published up to June 1971, have been well-documented by the U.S. Naval Medical Research Institute. Devices that emit RF and microwave radiation include, but is not limited to, cellphones, two-way radios, Wi-Fi routers, cellphone towers, smart watches, bluetooth devices, Smart meters, cordless cell phone base stations, wireless baby monitors, microwave ovens, and any WiFi-connected smart devices that receives and transmits data. Particular attention has been paid to the effects on man of non-ionizing radiation at these frequencies.
Reported Biological Phenomena (*Effects') and Some Clinical Manifestations Attributed to Microwave and Radio-Frequency Radiation A. Heating of Organs* (Applications: Diathermy, Electrosurgery, Electro-coagulation, Electrodesiccation, Electrotomy)
B. Changes in physiologic function
C. Central Nervous System Effects
D. Autonomic Nervous System Effects
E. Peripheral Nervous System Effects
F. Psychological Disorders ("Human Behavioral Studies") - the so-called "Psychophysiologic (and Psychosomatic) Responses"
G. Behavioral Changes (Animal)
H. Blood Disorders changes in:
I. Vascular Disorders
J. Enzyme and Other Biochemical Changes Changes in activity of:
K. Metabolic Disorders
L. Gastro-Intestinal Disorders
M. Endocrine Gland Changes
N. Histological Changes
O. Genetic and Chromosomal Changes
P. Pearl Chain Effect (Intracellular orientation of subcellular particles, and orientation of cellular and other (non-biologic) particles) Also, orientation of animals, birds, and fish in electromagnetic fields Q. Miscellaneous Effects
References
Glaser, Z. (1971). Bibliography of reported biological phenomena ('effects') and clinical manifestations attributed to microwave and radio-frequency radiation. Navel Medical Research Institute. https://archive.org/details/DTIC_AD0750271/mode/2up?view=theater
The study, conducted in the Fertility Clinic at Massachusetts General Hospital, is the first to examine associations between organophosphate flame retardants (PFRs)—which are used in polyurethane foam in many products, including upholstered furniture, baby products, gym mats, and even some phone cases—and reproductive outcomes in women. PFR's can be absorbed by the body through physical contact. These chemicals are not chemically bonded to foam and have been shown to migrate into the air and dust of indoor environments “These findings suggest that exposure to PFRs may be one of many risk factors for lower reproductive success,” said first author Courtney Carignan. “They also add to the body of evidence indicating a need to reduce the use of these flame retardants and identify safer alternatives.” Infertility, defined as the inability to get pregnant after 1 y or more of unprotected intercourse, affects one in six couples worldwide. One study found that pregnancy loss (miscarriage) affected approximately 28% of couples planning a pregnancy. Infertility has an associated health-care cost in the billions of dollars per year, not including the physical and psychological burden placed on the couple. Both the high rates of fertility along with the associated high costs should prompt the need to improve our understanding of risk factors that impair the ability to have a child. One potential risk factor is environmental exposure. Several classes of endocrine-disrupting chemicals (EDCs) with widespread general population exposure, including pesticides and phthalates, have been linked to infertility and adverse pregnancy outcomes. Since EDCs are ubiquitous, only a fraction have been evaluated for effects on infertility and pregnancy. PFRs are a class of EDCs with omnipresent exposure that have been detected in 90–100% of adult urine samples. Over the past decade, PFRs have been used widely in the polyurethane foam of upholstered furniture as replacements for pentabromodiphenyl ether, a flame retardant mixture that was phased out of use in 2005. Animal studies indicate that exposure to PFRs can disrupt endocrine function through altered thyroid action, steroidogenesis, or estrogen metabolism, and can also impair embryo development. Researchers have observed increasing PFR exposures were associated with a decrease in sperm motility and increased serum total T3 levels, in a small study of men. The aforementioned study explored the association between urinary concentrations of PFRs and pregnancy outcomes among women in a prospective cohort study, the Environment And Reproductive Health (EARTH) study, using assisted reproductive technologies (ART) as a model to study early developmental endpoints and pregnancy outcomes. In conclusion, using IVF (in vitro fertilization) as a model to investigate human reproduction and pregnancy outcomes, we found that concentrations of some urinary PFR metabolites were inversely associated with proportions of successful fertilization, implantation, clinical pregnancy, and live birth. These results highlight the potential reproductive effects of low-level exposure (i.e., background exposure levels of the general population) to PFRs and adverse IVF outcomes. Future research should focus on potential interactions between PFRs and other chemicals (i.e., mixtures analysis) that adversely affect reproductive health and also explore the potential effect of PFRs on male reproductive health. The work was supported by grants ES009718, ES022955, ES000002, and T32ES007069 from the National Institute of Environmental Health Sciences (NIEHS). References Carignan, C., Mínguez-Alarcón, L., Butt, C., Williams, P., Meeker, J., Stapleton, H., Toth, T., Ford, J. and Hauser, R. (2017). Urinary Concentrations of Organophosphate Flame Retardant Metabolites and Pregnancy Outcomes among Women Undergoing in Vitro Fertilization. Environmental Health Perspectives, 125(8). http://dx.doi.org/10.1289/EHP1021
Harvard. (2017). Common flame retardant chemicals may reduce likelihood of clinical pregnancy, live birth among women undergoing fertility treatments. [online] Available at: https://www.hsph.harvard.edu/news/press-releases/chemicals-flame-retardants-pregnancy/ [Accessed 31 Aug. 2017].
Ultraviolet radiation reaches the earth as UVA and UVB light, and has been classified as a human carcinogen by the National Toxicology Program (NTP) (National Toxicology Program, 2000). UVA is generally considered to be less carcinogenic than UVB. Originally, it was believed UVB light was more dangerous, thus sunscreen products were first developed to filter UVB and not UVA. However, recent research has demonstrated UVA radiation actually plays an important role in the development of malignant melanoma, the most aggressive form of skin cancer. Skin Cancer on the Rise - No Proof That Sunscreen Prevents Skin Cancer According to the National Cancer Institute, the rate of new melanoma cases among American adults has tripled since the 1970s, from 7.9 per 100,000 people in 1975 to 25.2 per 100,000 in 2014 (National Cancer Institute, 2017). A similar trend can be observed in regards to melanoma death rate for white American men, the highest risk group, which has escalated sharply, from 2.6 deaths per 100,000 in 1975 to 4.4 in 2014. From 2003 to 2012, the rates of new melanoma cases among both men and women have been increased by 1.7 and 1.4 percent per year (Centers for Disease Control and Prevention, 2016). While the exact cause of melanoma is unknown, researchers have established that risk factors for melanoma include family history, indoor tanning, the number of moles on a person’s skin, fair skin, freckles, blue or green eyes, blonde or red hair and a history of severe sunburns, among others (Centers for Disease Control and Prevention, 2017). People are able to modify only three of these risk factors: indoor tanning, exposure to UV radiation and severe sunburns. In December 2012, the Food and Drug Administration began to enforce new laws designed to improve sunscreens and consumer protection. The new laws restrict certain bogus label claims, but they allow most sunscreens to advertise “broad spectrum” skin protection. Sunscreen manufacturers are permitted to tell consumers, that with proper use, their products can help reduce the risk of skin cancer. However, the FDA and the International Agency for Research on Cancer have concluded that the available data does not support the assertion that sunscreens alone reduce the rate of skin cancer (Food and Drug Administration, 2011; IARC 2001). It's Not the Sunscreen, It's the Additives Recent research by the Center for Disease Control and Prevention found 96% of the U.S. population has oxybenzone in their bodies, a common chemical used in sunscreens; a chemical that is a known endocrine disruptor, linked to reduced sperm count in men and endometriosis in women (Environmental Working Group, 2017). Researchers have observed that adults who put on sunscreen containing 4% oxybenzone (the US allows up to 6%) in the morning and evening—mimicking what they’d do while on vacation—continued to excrete the chemical in their urine for five days afterwards, suggesting that it was being stored in the body (Gustavsson Gonzalez, Farbrot & Larko, 2002). Aside from oxybenzone — which is found in 70% of sunscreens — other commonly used chemicals that can enter your bloodstream and can cause toxic side effects, including hormone disruption, include but are not limited to:
Other chemicals, such as retinyl palmitate, may actually increase your risk of developing skin cancer. This product is a form of vitamin A that may speed the development of tumors and lesions when exposed to sunlight (National Toxicology Program, 2012). It is also advised to avoid using personal care products that contain synthetic fragrance, as this term describes any number of harmful chemicals that do not have to be listed individually on the label. Some common "fragrance" chemicals include:
Mineral Sunscreens May Contain Nanoparticles Mechanical sunscreens, including zinc oxide and titanium dioxide, have proven over years of use to be safe and effective mechanisms for blocking both UVA and UVB light. According to the Environmental Working Group (EWG) - a non-profit environmental organization that specializes in research and advocacy in the areas of toxic chemicals, agricultural subsidies, public lands, and corporate accountability - sunscreens made with zinc oxide and titanium dioxide are better alternatives because they provide strong sun protection with few health concerns and they don’t break down in the sun. Zinc oxide is EWG’s first choice for sun protection. Most studies to date have shown that zinc oxide and titanium dioxide are safe and unlikely to penetrate your skin when applied topically, as long as they are not nanosized. However, in an attempt to meet the desire of their consumers for products that don't leave a thick film on the skin, some manufacturers have reduced the size of the molecules, creating nanoparticles (microscopic particles measuring less than 100 nanometers). This nanotechnology has several different effects. Some are concerned that the particles have become so small that they may be absorbed directly into your skin. Although mixed, some studies have found significant negative health effects from the absorption of nanoparticles (European Union Public Health, 2006). While these nanoparticle technologies may make an excellent drug delivery system, it is questionable for use in sunscreen (De Jong & Borm, 2008). Titanium dioxide is more effective in UVB range and zinc oxide in the UVA range, therefore the combination of these particles assures a broad-band UV protection (Smijs & Pavel, 2011). Zinc oxide is beneficial because it remains stable in heat, but as a nanoparticle, the problems with toxicity probably outweigh the benefits to sun protection. Upon systemic distribution, toxicity of zinc oxide nanoparticles may affect the lungs, liver, kidneys, stomach, pancreas, spleen, heart and brain (Tian et al., 2015). Findings have also demonstrated that aging has a synergistic effect with zinc oxide nanoparticles on systemic inflammation and neurotoxicity, affecting your brain and neurological system. In other words, the older you are, the higher your risk of neurotoxicity from zinc oxide nanoparticle absorption. Spray-on sunscreens, containing zinc oxide and/or titanium dioxide, pose an additional hazard by releasing these toxic nanoparticles into the air. The FDA has previously expressed concern that inhaling these products may be risky, especially to children, and has warned parents to avoid spray-on sunscreens (Food and Drug Administration, 2006). While these two minerals are the safest topical sunscreen agents around, inhaling them is a whole different story. When these minerals are inhaled, they have been shown to irritate lung tissues and potentially lead to serious health problems, and the finer the particles, the worse their effects appear to be (Grassian, O’Shaughnessy, Adamcakova-Dodd, Pettibone & Thorne, 2006). The lungs have difficulty clearing small particles, and the particles may pass from the lungs into the bloodstream. Insoluble nanoparticles that penetrate skin or lung tissue can cause extensive organ damage. Some researchers speculate that the toxic effects of nanoparticles relate to their size being in the range of a virus, which may trigger your body's immune response (Buzea, Pacheco Blandino & Robbie, 2007). The International Agency for Research on Cancer (IARC) has classified titanium dioxide as a "possible carcinogen" when inhaled in high doses. According to IARC: "Titanium dioxide causes varying degrees of inflammation and associated pulmonary effects including lung epithelial cell injury, cholesterol granulomas and fibrosis. Rodents experience stronger pulmonary effects after exposure to ultrafine titanium dioxide particles compared with fine particles on a mass basis (IARC, 2006). The use of nanoparticles in cosmetics poses a regulatory challenge because the properties of nanoparticles may vary tremendously, depending on their size, shape, surface area and coatings. A number of manufacturers sell products advertised as containing “non-nano” zinc oxide and titanium dioxide - these claims are generally misleading. While particle sizes vary among manufacturers, nearly all would be considered nanomaterials under a broad definition of the term, including the definition proposed in 2011 by the Food and Drug Administration (Food and Drug Administration, 2011b). According to the available information, these mineral sunscreens must be delivered in nanoparticle form to render a layer that is reasonably transparent on the skin. According to EWG, even with the existing uncertainties, zinc oxide and titanium dioxide lotions are among the best choices on the American market. Here’s why:
Currently, all available evidence suggests that zinc oxide and titanium dioxide can be safely used in sunscreen lotions applied to healthy skin and pose a lower hazard than most other approved sunscreen ingredients. More human studies need to be conducted in regards to the health effects of inhaling of zinc oxide particles, especially at lower levels, such as from brief exposure to sunscreen spray. However, using these spray-on products are clearly an unnecessary risk since safer options are readily available. Your safest bet is to use topical zinc oxide or titanium dioxide that does not contain nanosized particles. High SPF is "Inherently Misleading" In theory, applying sunscreen with a sun protection factor (SPF) of 100 would allow sunbathers to be exposed to the sun 100 times longer before suffering a sunburn. For example, an individual who would normally redden after 30 minutes in the afternoon sun could theoretically stay out for 50 hours. But for high-SPF sunscreens, theory and reality are two different things. Researchers have observed that people are misled by the claims on high-SPF sunscreen bottles. They are more likely to use high-SPF products improperly and as a result may expose themselves to more harmful ultraviolet radiation than people relying on products with lower SPF values. The FDA has long contended that SPF higher than 50 is “inherently misleading” (Food and Drug Administration, 2007). Here are some reasons against applying sunscreens with SPF values greater than 50:
Researchers have conducted numerous studies on sunbathers and have observed that high-SPF products spur “profound changes in sun behavior” that may account for the increased melanoma risk found in some studies. The researchers confirmed that European vacationers spent more total time in the sun if they were given an SPF 30 sunscreen instead of an SPF 10 product (Autier et al., 2000). It is assumed the difference would also apply to products with SPF values greater than 50. Solutions: Sunscreen Should be the Last Resort You can boost your internal ability to offset UVA and UVB radiation through the consumption of specific nutrients each day. Antioxidants found in colorful fruits and veggies have been shown to have protective effects, but the omnipotent nutrient here is the fat-soluble carotenoid astaxanthin (asta-ZAN-thin), which is what gives krill, salmon, and flamingos their pink color (Ambati, Moi, Ravi & Aswathanarayana, 2014). Astaxanthin is produced by the microalgae Haematococcus pluvialis when its water supply dries up, forcing it to protect itself from ultraviolet radiation. It is this "radiation shield" mechanism that helps explain how astaxanthin can help protect you from similar radiation. When you consume this pigment, you are essentially creating your own "internal sunscreen." Researchers have confirmed astaxanthin is a potent UVB absorber that helps reduce DNA damage, reduce inflammation, oxidative stress and free radical damage throughout your body. Each of these health-promoting effects of astaxanthin improves the ability of your skin to handle sun without burning, while giving your body the best advantage to manufacturing vitamin D. However, it is still advised to seek physical protection from the sun, such as hat and long-sleeved clothing, but consuming more astaxanthin will provide a healthier alternative to using synthetic chemicals to filter UV radiation. Here are some helpful tips to help protect yourself from the sun's harmful UV rays:
When Purchasing Sunscreen Your safest and best choice for sunscreen protection is zinc oxide. However, avoid nanoparticles, which are common in spray sunscreens, to circumvent potential toxicity. Unfortunately, it can be challenging to find a product without other chemical additives. To help you choose the safest product, EWG performs an annual sunscreen evaluation based on effectiveness and safety. References Ambati, R., Moi, P., Ravi, S., & Aswathanarayana, R. (2014). Astaxanthin: Sources, Extraction, Stability, Biological Activities and Its Commercial Applications—A Review. Marine Drugs, 12(1), 128-152. http://dx.doi.org/10.3390/md12010128
Autier, P., Doré, J.-F., Reis, A. C., Grivegnée, A., Ollivaud, L., Truchetet, F., … Césarini, J.-P. (2000). Sunscreen use and intentional exposure to ultraviolet A and B radiation: a double blind randomized trial using personal dosimeters. British Journal of Cancer, 83(9), 1243–1248. http://doi.org/10.1054/bjoc.2000.1429 Buzea, C., Pacheco Blandino, I., & Robbie, K. (2007). Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases. Retrieved 3 August 2017, from https://arxiv.org/ftp/arxiv/papers/0801/0801.3280.pdf Centers for Disease Control and Prevention. (2017). What Are the Risk Factors for Skin Cancer?. Cdc.gov. Retrieved 3 August 2017, from https://www.cdc.gov/cancer/skin/basic_info/risk_factors.htm Centers for Disease Control and Prevention. (2016). Skin Cancer Trends. (2017). Cdc.gov. Retrieved 3 August 2017, from https://www.cdc.gov/cancer/skin/statistics/trends.htm De Jong, W. H., & Borm, P. J. (2008). Drug delivery and nanoparticles: Applications and hazards. International Journal of Nanomedicine, 3(2), 133–149. Environmental Working Group. (2017). EWG's 2017 Guide to Safer Sunscreens. Ewg.org. Retrieved 3 August 2017, from http://www.ewg.org/sunscreen/report/the-trouble-with-sunscreen-chemicals/#.WYKSS4UnHPp European Union Public Health. (2006). What are potential harmful effects of nanoparticles?. Ec.europa.eu. Retrieved 3 August 2017, from http://ec.europa.eu/health/scientific_committees/opinions_layman/en/nanotechnologies/l-3/6-health-effects-nanoparticles.htm Food and Drug Administration. (2011). Labeling and Effectiveness Testing: Sunscreen Drug Products for Over-the-Counter Human Use. Regulations.gov. Retrieved 3 August 2017, from https://www.regulations.gov/document?D=FDA-1978-N-0018-0698 Food and Drug Administration. (2011b). FDA Draft, Not for Implementation: Guidance for Industry. Enforcement Policy – OTC Sunscreen Drug Products Marketed Without an Approved Application. Available at www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM259001.pdf Food and Drug Administration. (2006). PETITION REQUESTING FDA AMEND ITS REGULATIONS FOR PRODUCTS COMPOSED OF ENGINEERED NANOPARTICLES GENERALLY AND SUNSCREEN DRUG PRODUCTS COMPOSED OF ENGINEERED NANOPARTICLES SPECIFICALLY. FDA.gov. Retrieved 3 August 2017, from https://www.fda.gov/ohrms/dockets/dockets/06p0210/06p-0210-cp00001-01-vol1.pdf Food and Drug Administration. (2007). Sunscreen Drug Products for Over-the-Counter Human Use; Proposed Amendment of Final Monograph; Proposed Rule - FR Doc 07-4131. (2017). Fda.gov. Retrieved 3 August 2017, from https://www.fda.gov/OHRMS/DOCKETS/98fr/07-4131.htm Grassian, V., O’Shaughnessy, P., Adamcakova-Dodd, A., Pettibone, J., & Thorne, P. (2006). Inhalation Exposure Study of Titanium Dioxide Nanoparticles with a Primary Particle Size of 2 to 5 nm. Environmental Health Perspectives, 115(3), 397-402. http://dx.doi.org/10.1289/ehp.9469 Gustavsson Gonzalez, H., Farbrot, A., & Larko, O. (2002). Percutaneous absorption of benzophenone-3, a common component of topical sunscreens. Clinical And Experimental Dermatology, 27(8), 691-694. http://dx.doi.org/10.1046/j.1365-2230.2002.01095.x International Agency for Research on Cancer (IARC). (2006). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. IARC.fr. Retrieved 3 August 2017, from https://monographs.iarc.fr/ENG/Monographs/vol93/mono93.pdf IARC. (2001). IARC Summary Recommendations for Public Health Action. IARC. Retrieved 3 August 2017, from http://www.who.int/uv/resources/recommendations/en/IARCSum.pdf Mercola, J. (2017). Is Your Sunscreen Doing More Harm Than Good?. Mercola.com. Retrieved 3 August 2017, from http://articles.mercola.com/sites/articles/archive/2017/04/26/hazardous-chemicals-in-sunscreens.aspx National Cancer Institute. (2017). Melanoma of the Skin - Cancer Stat Facts. Seer.cancer.gov. Retrieved 3 August 2017, from http://seer.cancer.gov/statfacts/html/melan.html National Toxicology Program (NTP). (2000). Broad-Spectrum Ultraviolet (UV) Radiation and UVA, and UVB, and UVC. U.S. Department of Health and Human Services. Retrieved 3 August 2017, from https://ntp.niehs.nih.gov/ntp/newhomeroc/roc10/uv_no_appendices_508.pdf NTP Technical Report on the Photococarcinogenesis Study of Retinoic Acid and Retinyl Palmitate [CAS Nos. 302-79-4 (All-Trans-Retinoic Acid) and 79-81-2 (All-Trans-Retinyl Palmitate)] in SKH-1 Mice (Simulated Solar Light And Topical Application Study). NTP TR 568, National Institutes of Health, 2012. Available at ntp.niehs.nih.gov/ntp/htdocs/LT_rpts/TR568_508.pdf Popov, A., Haag, S., Meinke, M., Lademann, J., Priezzhev, A., & Myllylä, R. (2009). Effect of size of TiO[sub 2] nanoparticles applied onto glass slide and porcine skin on generation of free radicals under ultraviolet irradiation. Journal Of Biomedical Optics, 14(2), 021011. http://dx.doi.org/10.1117/1.3078802 SCCNFP, Opinion Concerning Titanium Dioxide. Opinion: European Commission – The Scientific Committee on Cosmetic Products and Non-Food Products Intended for Consumers. (2000). Retrieved 3 August 2017, from http://ec.europa.eu/health/ph_risk/committees/sccp/documents/out135_en.pdf Scientific Committee on Consumer Safety. (2012). Opinion on Zinc oxide (nano form). Retrieved 3 August 2017, from http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_103.pdf Smijs, T. G., & Pavel, S. (2011). Titanium dioxide and zinc oxide nanoparticles in sunscreens: focus on their safety and effectiveness. Nanotechnology, Science and Applications, 4, 95–112. http://doi.org/10.2147/NSA.S19419 Tian, L., Lin, B., Wu, L., Li, K., Liu, H., & Yan, J. et al. (2015). Neurotoxicity induced by zinc oxide nanoparticles: age-related differences and interaction. Scientific Reports, 5(1). http://dx.doi.org/10.1038/srep16117 |
The Awareness domain contains research, news, information, observations, and ideas at the level of self in an effort to intellectualize health concepts.
The Lifestyle domain builds off intellectual concepts and offers practical applications.
Taking care of yourself is at the core of the other domains because the others depend on your health and wellness.
Archives
May 2024
Categories
All
|




 RSS Feed
RSS Feed

