|
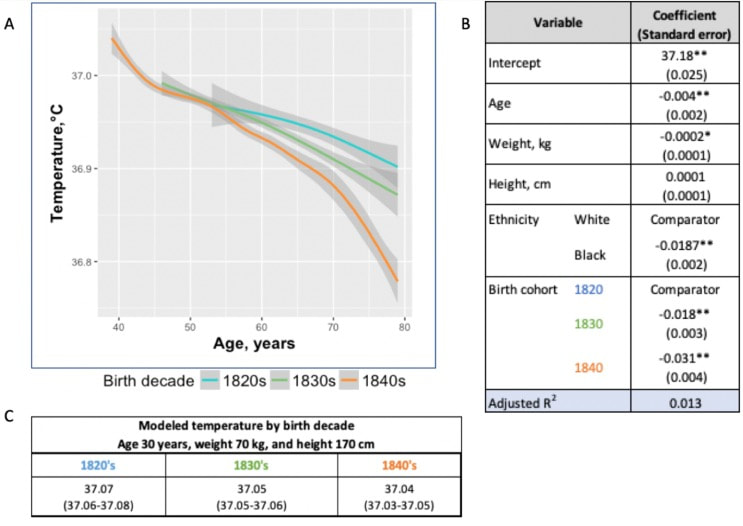
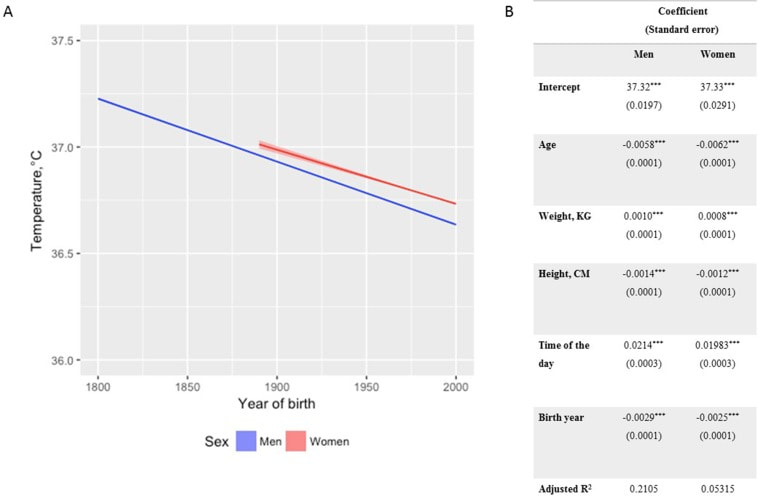
In recent years, researchers have uncovered a surprising trend in human physiology: a decline in body temperature over the past two centuries. Contrary to the long-standing belief that the normal body temperature is 37°C (98.6°F), studies spanning multiple cohorts and time periods have revealed a consistent decrease in average body temperature, suggesting a real physiological change rather than a mere artifact of measurement bias. A groundbreaking study analyzed data from three cohorts spanning 157 years (over 600,000 data inputs), including Union Army Veterans of the Civil War, the National Health and Nutrition Examination Survey I, and the Stanford Translational Research Integrated Database Environment. The findings revealed a monotonic decrease in body temperature, with men born in the early 19th century having temperatures 0.59°C higher than men today. A similar decline was observed in women, indicating a significant shift in human physiology over time. While some may attribute these findings to changes in measurement methods or biases, the study's authors argue that the observed drop in temperature reflects real physiological differences. Human body temperature is a crude surrogate for basal metabolic rate. These findings of a decrease in body temperature indicate a decrease in metabolic rate, which is supported in the literature when comparing modern experimental data to those from 1919. Resting metabolic rate, a key component of daily energy expenditure, has been linked to body temperature and longevity. The observed decrease in body temperature suggests a corresponding decline in metabolic rate, independent of changes in body size. This likely has implications for human health and longevity, as metabolic health underlies all vital organ functions. Additionally, changes in ambient temperature and the widespread adoption of heating and cooling systems in modern times may have influenced body temperature trends. Increased time spent in thermoneutral zones, where minimal energy is expended to maintain body temperature, could contribute to the observed decline in resting metabolic rate and body temperature. In conclusion, body temperature has declined, implying lower metabolic rates (since heat is generated as a byproduct of energy production). This declining trend in human body temperature over the past two centuries offers valuable insights into the complex interplay between physiology, environment, and health. As researchers continue to unravel the mysteries of human biology, these findings pave the way for a deeper understanding of our species' evolution and resilience in the face of changing environments and lifestyles. referencesProtsiv, Myroslava, et al. “Decreasing Human Body Temperature in the United States since the Industrial Revolution.” ELife, vol. 9, 7 Jan. 2020, p. e49555, elifesciences.org/articles/49555, https://doi.org/10.7554/eLife.49555.
Dai, Dao-Fu, et al. “Mitochondrial Oxidative Stress in Aging and Healthspan.” Longevity & Healthspan, vol. 3, no. 1, 2014, p. 6, https://doi.org/10.1186/2046-2395-3-6.
2 Comments
|
The Awareness domain contains research, news, information, observations, and ideas at the level of self in an effort to intellectualize health concepts.
The Lifestyle domain builds off intellectual concepts and offers practical applications.
Taking care of yourself is at the core of the other domains because the others depend on your health and wellness.
Archives
May 2024
Categories
All
|
 RSS Feed
RSS Feed

