|
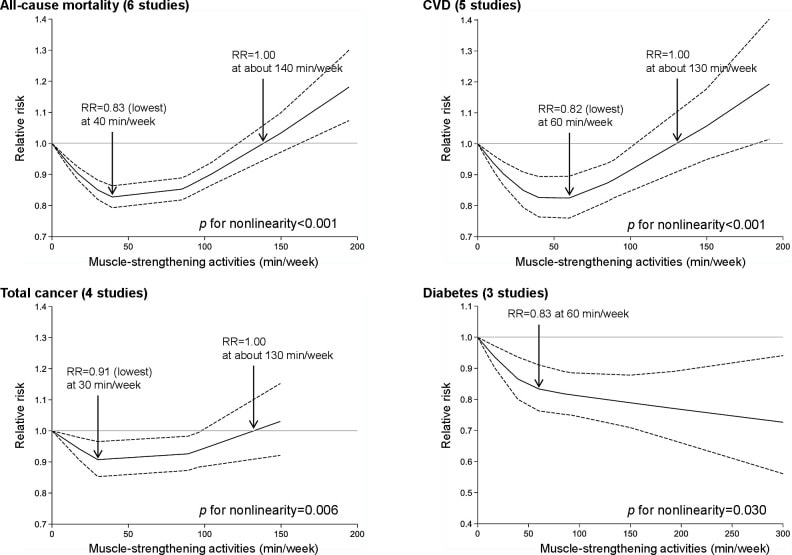
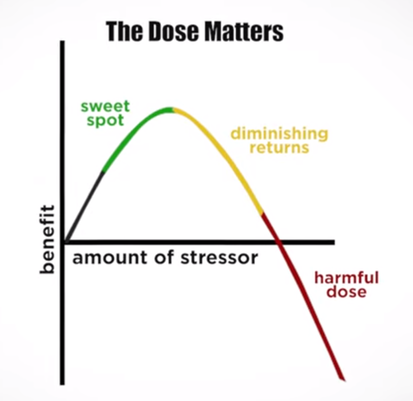
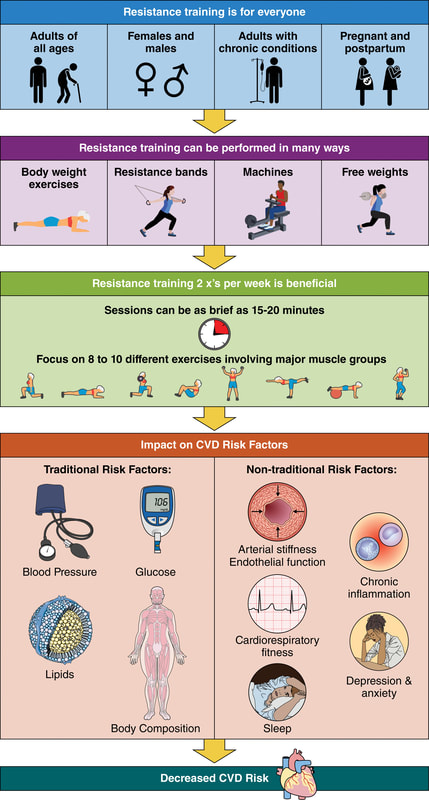
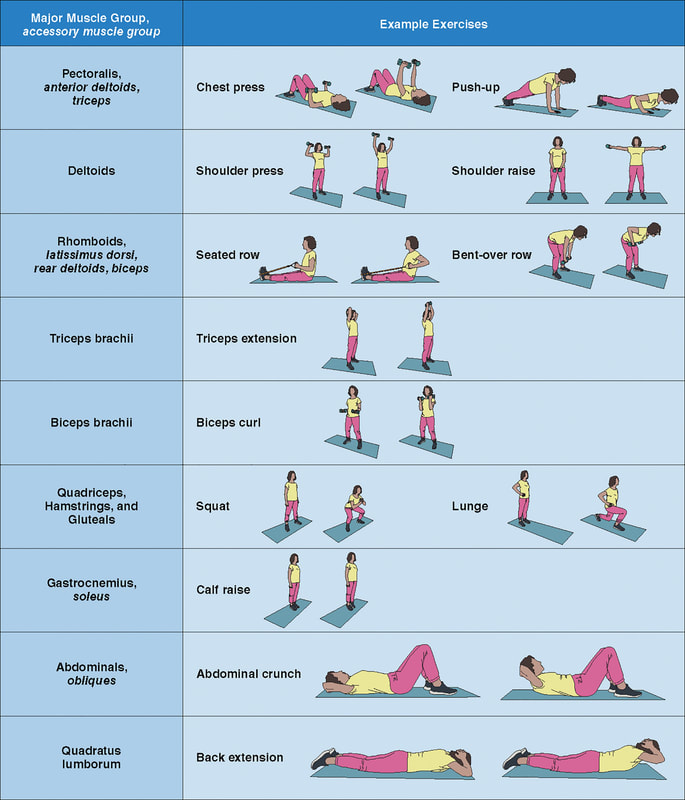
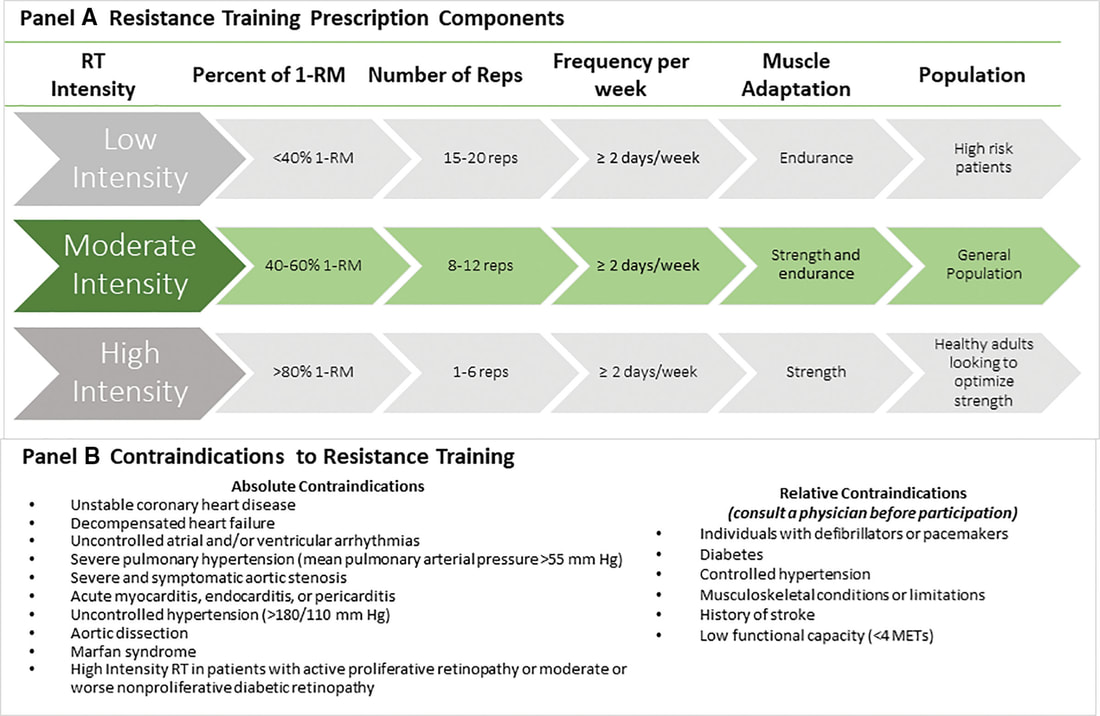
In an update to its 2007 scientific statement, the American Heart Association (AHA) emphasizes the significant and multifaceted benefits of resistance training (RT) on cardiovascular health. Contrary to the misconception that RT solely enhances muscle mass and strength, the statement highlights the favorable physiological and clinical effects of this form of exercise on cardiovascular disease (CVD) and associated risk factors. The scientific statement aims to provide comprehensive insights into the impact of RT, either alone or in combination with aerobic training, on traditional and nontraditional CVD risk factors. More is not always betterEpidemiological evidence suggests that RT is associated with a lower risk of all-cause mortality and CVD morbidity and mortality. Adults who participate in RT have ≈15% lower risk of all-cause mortality and 17% lower risk of CVD, compared with adults who report no RT. Approximately 30 to 60 minutes per week of RT is associated with the maximum risk reduction for all-cause mortality and incident CVD. Notice this "U" shape in the curve when examining the relationship between RT and morbidity and mortality. This curve suggests that some RT is clearly beneficial, but has the volume of RT increases past a certain point the benefits drop and it becomes harmful. The concept of a "biphasic response" is fundamental to understanding hormesis. It describes the characteristic dose-response relationship observed in hormetic processes, where a substance or stressor elicits opposite effects at low and high doses. The response can be visualized as a U-shaped or J-shaped curve, illustrating the beneficial effects at low doses and potential harm at higher doses. Benefits of RT on Traditional CVD Risk FactorsThe AHA's scientific statement underscores the positive influence of RT on traditional CVD risk factors, including blood pressure (BP), glycemia, lipid profiles, and body composition. Numerous studies indicate that engaging in RT is associated with reduced resting BP, improved glycemic control, and favorable alterations in lipid profiles, contributing to a lower risk of all-cause mortality and CVD morbidity. Despite recommendations suggesting 2 days per week of RT, only 28% of U.S. adults adhere to this guideline, highlighting the need for increased awareness and promotion. RT and resting blood pressureRT has demonstrated the ability to reduce resting BP across diverse populations, with notable benefits observed in individuals with prehypertension and hypertension. The mechanisms behind these benefits include enhancements in endothelial function, vasodilatory capacity, and vascular conductance. The reductions in BP achieved through RT are comparable to those achieved with antihypertensive medications. RT and GlycemiaRT shows promise in improving glycemia and insulin resistance, leading to a lower incidence of diabetes. The evidence suggests a nonlinear dose-response association, with up to 60 minutes per week of RT associated with the maximum risk reduction for diabetes. RT and Lipid ProfilesWhile the effect on lipid profiles is modest, RT results in favorable changes in high-density lipoprotein cholesterol, total cholesterol, and triglycerides. These improvements are more pronounced in older adults and those with elevated cardiometabolic risk. Rt, Body composition, and weightRT positively influences body composition by increasing lean body mass and reducing body fat percentage. It is particularly effective in overweight or obese individuals, contributing to increased metabolic rate and mitigating weight gain over time. Benefits of RT on Nontraditional CVD Risk FactorsIn addition to traditional risk factors, the scientific statement highlights the potential mechanisms by which RT positively affects nontraditional CVD risk factors. These include increased cardiorespiratory fitness, improved endothelial function, and potential benefits for sleep quality, psychological health, and well-being. The AHA's updated scientific statement reinforces the pivotal role of resistance training in cardiovascular health, providing a comprehensive overview of its impact on both traditional and nontraditional risk factors. As the evidence supporting RT's benefits continues to grow, the statement serves as a valuable resource for clinicians and public health professionals, offering practical strategies for promoting and prescribing resistance training to enhance cardiovascular health in diverse populations. ReferencesPaluch, Amanda E, et al. “Resistance Exercise Training in Individuals with and without Cardiovascular Disease: 2023 Update: A Scientific Statement from the American Heart Association.” Circulation, 7 Dec. 2023, https://doi.org/10.1161/cir.0000000000001189. Accessed 11 Dec. 2023.
Momma H, Kawakami R, Honda T, Sawada SS. Muscle-strengthening activities are associated with lower risk and mortality in major non-communicable diseases: a systematic review and meta-analysis of cohort studies. Br J Sports Med. 2022 Jul;56(13):755-763. doi: 10.1136/bjsports-2021-105061. Epub 2022 Feb 28. PMID: 35228201; PMCID: PMC9209691.
1 Comment
We’ve built an entire economy, not just in the United States but the entire Western civilization, on healthcare. For thousands of years, real control over populations has been around their food. Today, with billions of souls on the planet, controlling food has become a massive business and a means of ultimate political control. Between 1982 and 2000, something changed in our environment, overwhelming the immune system of the population. Diseases in different organ systems started going epidemic simultaneously, challenging the notion of a thousand different diseases.
In the late 1800s, we changed the way we farmed, leading to a disrespect for crop rotation and soil health. This disrespect for soil health resulted in the Dust Bowl of the 1920s and 30s, pushing us to outsource our food production and rely on imported food. After World War II, with a surplus of petroleum, we started producing chemical-based fertilizers, leading to the Green Revolution. While plants turned green due to nitrogen and phosphorus, they lacked essential nutrients and medicine. This deficiency weakened plants, making them susceptible to diseases and pests. The chemical industry introduced pesticides and herbicides (including #Glyphosate), creating a co-dependent relationship between farmers and chemical solutions. Similarly, in healthcare, we’ve become dependent on drugs to manage symptoms, creating a cycle of side effects and more medications. The epidemic rise in diseases like autism, Alzheimer’s, Parkinson’s, and autoimmune disorders signals a deeper problem, challenging our understanding of the root cause of diseases. It’s time to reconsider our approach to health, starting with understanding the importance of soil health and nutrition. Just as respecting soil is crucial for healthy crops, prioritizing our body’s nutritional needs is fundamental for overall well-being. Let’s shift our focus from symptom management to addressing the root cause, promoting a holistic approach to health. Zach Bush, MD is triple board-certified physician specializing in internal medicine, endocrinology and hospice care. He is the founder of Seraphic Group, an organization devoted to developing root-cause solutions for human and ecological health in the sectors of big farming, big pharma, and Western Medicine at large. And he is also the founder of Farmers Footprint https://farmersfootprint.us/, a non-profit coalition of farmers, educators, doctors, scientists, and business leaders aiming to expose the deleterious human and environmental impacts of chemical farming and pesticide reliance - while simultaneously offering a path forward through regenerative agricultural practices. Zach Bush MD is a physician specializing in internal medicine, endocrinology and hospice care. He is an internationally recognized educator and thought leader on the microbiome as it relates to health, disease, and food systems. Dr Zach founded Seraphic Group and the nonprofit Farmer’s Footprint to develop root-cause solutions for human and ecological health. His passion for education reaches across many disciplines, including topics such as the role of soil and water ecosystems in human genomics, immunity, and gut/brain health. His education has highlighted the need for a radical departure from chemical farming and pharmacy, and his ongoing efforts are providing a path for consumers, farmers, and mega-industries to work together for a healthy future for people and planet.
Show Notes
From RT:
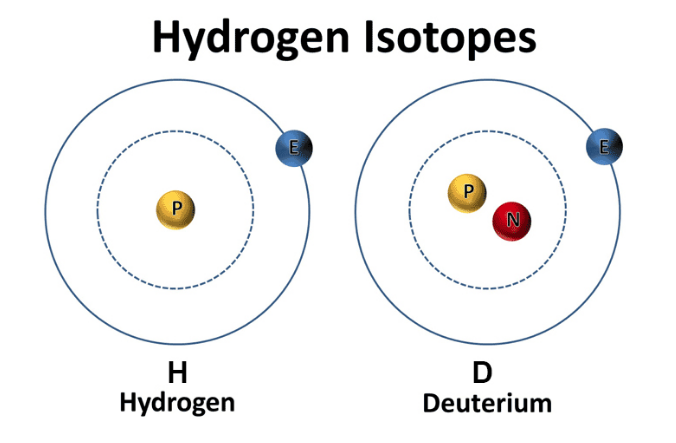
Now, we have a thing called a messenger RNA vaccine (mRNA). RNA is, effectively, a single strand of DNA – the double helix that sits within our cells and makes up our genetic code. Many viruses are made up of a single strand of RNA, surrounded by a protein sphere. They enter the cell, take over the replication systems, make thousands of copies of themselves, then exit the cell. Sometimes killing the cell as they do so, sometimes exiting more gently. Covid19 (Sars-Cov2) is an RNA virus. Knowing this, rather than attempting to create a weakened virus, which can take years, or break the virus into bits, the vaccine researchers decided to use Sars-Cov2’s RNA against itself. To do this, they isolated the section of RNA which codes for the ‘spike’ protein – which is the thing the virus uses as a ‘key’ to enter cells. They then worked out how to insert this small section of RNA, messenger RNA, into the cell, where it takes over a part of the protein replication mechanisms that sit inside all cells. They turn the mechanism into a 3D printer, churning out copies of the spike protein. These spike proteins then leave the cell – somehow or other, this bit is unclear. The immune system comes across them, recognises them as ‘alien’ and attacks. In doing so, antibodies are created, and the immune memory system kicks into action. If, later on, a Sars-Cov2 virus gets into the body, the immune system fires up and attacks the remembered spike protein. Hopefully killing the entire virus. This is all, certainly very clever stuff. What, as they say, could possibly go wrong? The first thing to say is that, with something this new, we don’t really know. It could be that it is absolutely 100 percent safe. We are told that none of the mRNA can get into the nucleus of the cell, where it could become incorporated into the DNA. I hope so. Could it trigger an immune cascade? I hope not. I know that the researchers will be looking very, very, closely at the novel safety issues that could emerge. If they are not, they damned well should be. However, the timelines here are very short. It normally takes many years to create safe and effective vaccines. Here is it happening in, effectively, weeks. Hydrogen comes in two “flavors”: regular hydrogen, which is actually called protium, and deuterium. Deuterium has all of the same properties as hydrogen, except that it's twice as big and heavy. This is due to an added neutron paired with the proton in the nucleus. Because of this, deuterium is also referred to as "heavy hydrogen," and it actually behaves quite differently from regular hydrogen in chemical reactions and in our bodies. In nature, deuterium helps things grow. For example, deuterium is biologically necessary for growth in babies, teenagers, and developing plants and animals. But once you stop growing, having too much deuterium in your cells can result in mitochondrial dysfunction and lead to premature aging, metabolic problems, and disease. Deuterium is like thick, gluggy oil - when you put thick oil into an engine, the engine sputters, makes strange noises, and eventually breaks. Nature has put systems in place to deplete deuterium and protect the nanomotors, or "little engines," in our cells’ mitochondria from coming into contact with this thick oil. However, the side effects of a modern life - pollution, global warming, processed foods, less healthy lifestyles, etc. - have resulted in many people having way too much deuterium inside their cells. This results in an inability to effectively deplete deuterium and the destruction of our nanomotors. This starts a vicious cycle of deuterium building up and breaking more of our nanomotors. Fewer nanomotors means less energy and more sickness and disease. While deuterium is a natural and essential element, its presence has increased in the modernized environment within the food, atmosphere, and water. Deuterium levels is food will vary based o where that food is grown - deuterium is highest in the equator and in low elevations. Foods high in fat, as well as green plants, including algae and spirulina, which contain high amounts of chlorophyll, are lower in deuterium than fruits, roots, and underground vegetables. As it turns out, GMO foods tainted with glyphosate, as well as processed, synthetically made foods, possess high amounts of deuterium. There are various lifestyle practices that have led to increased deuterium levels including a lack of sleep, particularly deep REM sleep. In addition, breathing shallow and fast via the mouth and chest also contributes to elevated levels of deuterium. Researchers have demonstrated that elevated of deuterium can contribute to:
Learn More About Deuterium Depletion References Understanding Deuterium - The Center for Deuterium Depletion. (2019). Retrieved 24 December 2019, from https://www.ddcenters.com/about-deuterium-2-2-2/
It is widely recognized that radiation exposures such as X-rays and gamma radiation can increase the risk of cancer in humans and animals. These types of radiation are referred to as ionizing radiation (Ionization energy is defined as the minimum amount of energy required to remove an electron from an atom or molecule in the gaseous state). This is different from nonionizing radiation, which includes ultraviolet (UV), visible light, extremely low frequency radiation (ELF), and radiofrequency or microwave (RF) radiation. Conventionally, researchers believed that nonionizing radiation is not harmful or carcinogenic, despite evidence surfacing regarding the relationship between UV radiation and skin cancer. Research evaluating the exposure to RF radiation was conducted primarily by military agencies. Due to the advent and use of cellular telephone systems, which involve widespread public exposures, reevaluation of exposure risk has become urgent. Four types of physiological effects has been observed by researched in multiple studies:
These findings suggest that exposure to RF radiation, including from devices such as microwave ovens, are potentially carcinogenic and have other health effects. An important point to consider is that low dose radiation exposure over time has damaging effects, rather than one large exposure to radiation. Alternatives to Microwaves Rather than using a microwave, consider using a stove or convection oven. These methods take a bit more time, but it is well worth it! Consider using headphones to talk on cell phones rather than placing the device directly to your head. If possible, request to avoid airport scanners by opting for a pat-down. Lastly, try to reduce overall radiation exposure over time. References Goldsmith, J. (1997). Epidemiologic Evidence Relevant to Radar (Microwave) Effects. Environmental Health Perspectives, 105, 1579. https://doi.org/10.2307/3433674
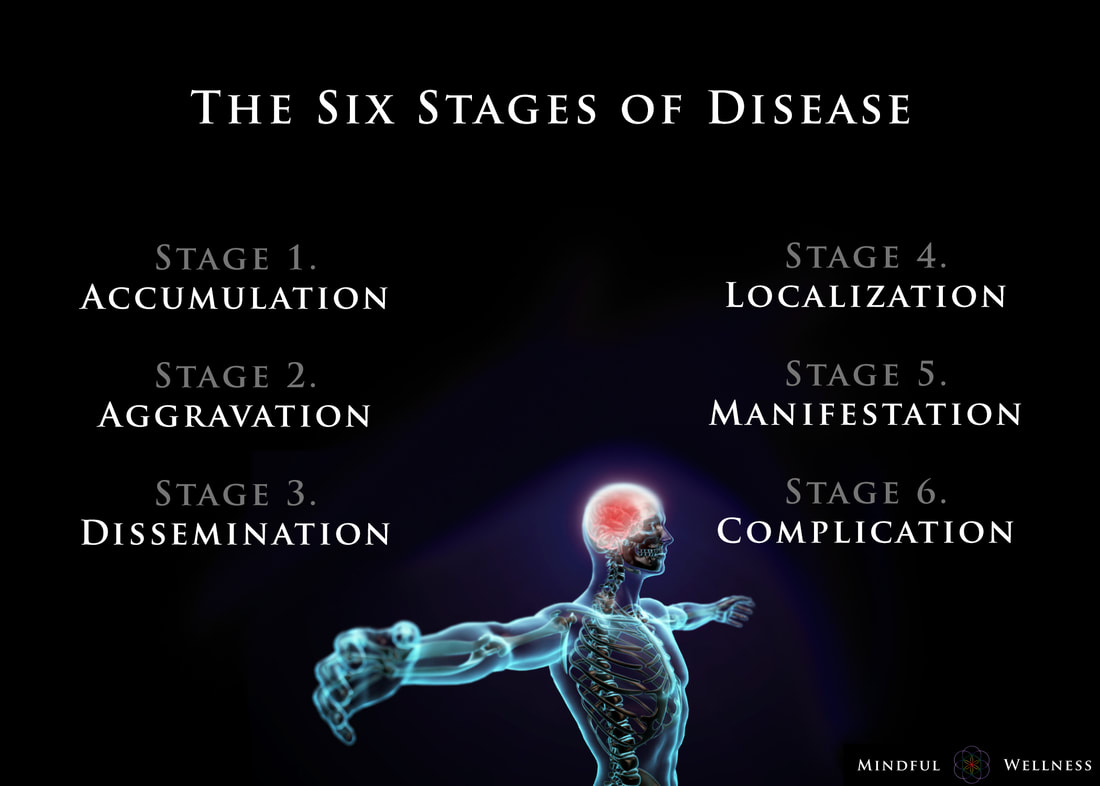
Ayurveda offers insight into the earlier stages and enables those monitoring their health to take care of any small imbalances well before developing any serious illness. The length of the each stage may vary from weeks, to months, even years, depending on the person and the degree of aggravation. The six stages of disease development are:
1. Accumulation The first stage, accumulation, represents imbalance, a build up or collection of something in the body. Being exposed to and acquiring a pathogen via the external environment is an example of accumulation. This stage can also be caused by the internal environment, such as from eating an imbalanced diet leading to excess inflammation or mucous. Accumulation in the body leads to the the next stage, aggravation. 2. Aggravation As the imbalanced elements continue to increase, the symptoms become more aggravated and will begin to be noticed throughout the body. This stage is a sign of continued accumulation. This stage can manifest, as seen in the Kapha state, as loss of appetite, indigestion, nausea, excess saliva, oversleeping, sluggishness; or as seen in the Pitta state, as increased acidity, burning sensations in the abdomen, lowered vitality, or insomnia; or as seen in the Vata state, as pain in the lower abdomen, excess flatulence, and light-headedness. 3. dissemination Once the site of origin is full with excess accumulation and is aggravated, it will begin to overflow into or disseminate throughout the rest of the body using different channels of transportation. Overflow typically begins in the GI tract, then spilling into the circulating plasma and blood, allowing the accumulation to spread systemically, and eventually seeping into the organs and tissues (dhātus). Simultaneously, the symptoms at the site of origin will grow worse. 4. Localization The excess accumulation will then move to wherever a weak site exists in the body. This is where and when diseases begin to develop. This stage is also where genetics matter; the weak spots are determined by genetics - as the saying goes, genetics loads the gun, environment pulls the trigger. This stage can manifest, as seen in the Vata state, as arthritis. In a Pitta state, this can be seen as an ulcer, and in the Kapha state, manifestation may begin in the lungs. At this stage, healing is still regarded as simple. 5. Manifestation This is the first state of the development of illness for which modern Western medicine can detect signs of disease. It is at this stage where diseases progress and become fully developed, showing signs of clinical features. Manifested imbalances are given names at this stage, such as arthrosclerosis, cancer, colitis, etc. It is at this stage where conventional medicine attempts to mask the symptoms by offering pharmaceutical drugs. 6. Complication Complications of the dis-ease begin at this final stage. Often times, conventional medicine attempts to solve the problem by simply removing the affected tissue (e.g., small intenstine, colon, thyroid, etc.) from the body. The symptoms become clear enough so that the elemental cause (i.e., dosha constitution such as Vata, Kapha, Pitta) may be determined. Some medical professionals describe this stage as the chronic phase of development. For example, if one develops inflammation in the manifestation stage, in this stage, complications set in, and the inflammation may grow worse into a chronic problem. Being aware of the stages of the dis-ease process is helpful because one can gain a better understanding in how prevent, and perhaps even reverse, it. To be clear, the information provided here is not claiming to treat, cure or diagnose disease. ReferencesCabral, S. (2018). The 6 Stages of the Disease Process (Ayurvedic Principle). [podcast] The Cabral Concept. Available at: https://itunes.apple.com/us/podcast/cabral-concept-by-stephen/id1071469441?mt=2
Tirtha, S. (2007). The Āyuveda encyclopedia. Bayville, NY. Ayurveda Holistic Center Press. The environment that we live in is toxic. It is worrisome to think that the status quo has occurred with the help of corporations knowingly dumping harmful chemicals into the environment.
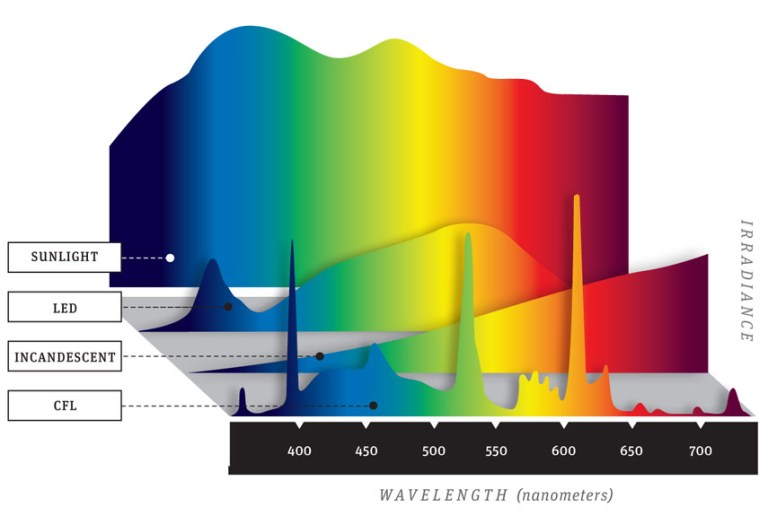
The Environmental Working Group (EWG) has studied the current state of our world in great detail and has discovered that before a child is even born they already have approximately 287 toxins in their blood and tissues. These results came from 10 newborns whose parents gave permission to have their toxins measured at birth. The results of this study indicate that an average of 200 chemicals was found in each newborn. Of the toxins tested, 47 were consumer ingredients such as cosmetics, 212 were industrial and pesticide byproducts. In this study, only around 400 total chemicals were actually tested for - thousands of others may have been found if larger parameters were used. Many of the toxins measured in the newborns included plastics, flame retardants, and other chemicals that disrupt brain function, IQ, hormones, and the nervous system of the child. Some of the toxins observed like DDT, have actually been banned since 1972 (over 3 decades ago), but are still being measured in laboratory samples. Certain chemicals never fully degrade in the environment. So, there is not question about it, the environment we live in is toxic. All of us have disease-creating toxins inside of our bodies, the question boils down to which ones and how much. But there are some solutions: lab tests and detoxification. Transcript: "You hear all this talk about blue light being harmful, but do you know what blue light actually is? Blue light is one of the types of light that form the white light we get from the Sun. Together with red, orange, yellow, green, violet, and indigo. This is called the electromagnetic (EM) spectrum of visible light. At the end we have the UV light, which can't actually be seen by the human eye. The energy of these waves increases as we go towards the end, which makes blue lights one of the highest intensity types of visible light. Light is made of EM waves that emit energy and it's this energy that we perceive as light. These waves come in different wavelengths, which means we get different colors of light. These are the different colors of light that we can perceive from the EM spectrum. So blue light is actually everywhere; it's in the light that travels from the Sun all the way to the Earth. Because the wavelength of blue light is so small they collide with air molecules a lot more than any other color and they get scattered everywhere - that's actually what makes the sky blue. That's why your body uses this blue light from the Sun to make the difference between day and night and regulate your sleep cycle. But our eyes natural filters barely provide us with enough protection from blue light on a particularly sunny day. The blue light from your devices is even worse. LED devices emit much stronger blue light than we get from the Sun. Spending hours staring at a screen can cause eye damage and fatigue. This is due to most lower energy waves being absorbed by the cornea, the eyes outer membrane. Blue light goes straight through due to its high energy and slowly deteriorates the retina. And because our brains use blue light to differentiate between day and night and boost alertness, spending time on your phone tablet or laptop late at night fools your body into thinking it should keep you awake. It's all of them - phones, tablets, any gadget with an illuminated display. They all use blue LEDs because they are energy efficient and cheaper to produce, but your body disagrees. Basically, what keeps you awake and alert during the day can severely affect the quality of your sleep at night. Blue light has also been shown to suppress secretion of melatonin, a hormone that is produced at night and helps your body prepare for sleep. Melatonin isn't just linked to poor
sleep, scientists have also managed to find a correlation between melatonin deprivation and conditions like cancer, diabetes and clinical depression. Do you still think your tablet is that harmless? Well there is a way you can prevent it. Scientists have now designed special screen protectors that stop the blue light from reaching your eyes and causing damage to your retina at a microscopic level. This glass has tiny ridges that block blue waves and let the other less harmful light go through. These glasses can block up to 60% of blue light and 99% of UV rays. People have reported that their sleeping patterns were significantly improved after only a few days. There was also significant reduction in eye strain and headaches. Or you can go for full protection and buy eyewear that you can use for all blue light-emitting devices: computers, TV laptops, or your phone. These work in a slightly different way - the protectors as the yellow, absorbs rather than blocks blue and UV light, and lets other types of light go through. No matter what you do, whether its buying a protective glass or reducing the time you spend on your device make sure you stay on the lookout for the unseen damage that your day-to-day gadgets can do to your health." Welcome to the first episode of our brand new docuseries - Remedy: Ancient Medicine for Modern Illness.
(If you are not registered for the full Remedy docuseries yet, click this link to join us for all 9 episodes - https://remedy.thesacredscience.com/r... ) Episode 1 is called “The Quest For Lost Medicine” and it lays the groundwork for the entire series - some of what you will witness may shock you… Over the 9-part Remedy series, we’ll be uncovering powerful herbal remedies for major diseases - but first, we need to understand why this vital healing information has been kept from us. Here’s some of what we will reveal in this first episode…
This eye-opening episode sets the stage for everything else we cover in the Remedy docuseries. Again, if you are not registered for the full series, click here - https://remedy.thesacredscience.com/r... Top Cancer Researcher Fails to Disclose Corporate Financial Ties in Major Research Journals9/8/2018
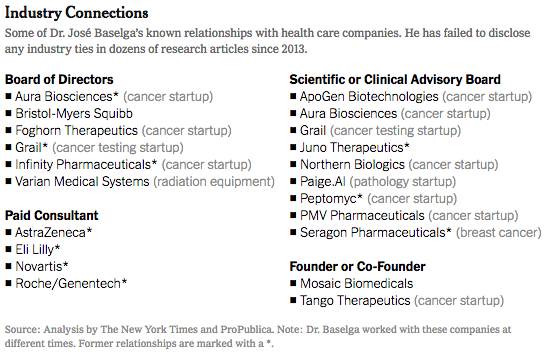
The researcher, Dr. José Baselga, a towering figure in the cancer world, is the chief medical officer at Memorial Sloan Kettering Cancer Center in New York. He has held board memberships or advisory roles with Roche and Bristol-Myers Squibb, among other corporations, has had a stake in start-ups testing cancer therapies, and played a key role in the development of breakthrough drugs that have revolutionized treatments for breast cancer. According to an analysis by The New York Times and ProPublica, Dr. Baselga did not follow financial disclosure rules set by the American Association for Cancer Research when he was president of the group. He also left out payments he received from companies connected to cancer research in his articles published in the group’s journal, Cancer Discovery. At the same time, he has been one of the journal’s two editors in chief. At a conference this year and before analysts in 2017, he put a positive spin on the results of two Roche-sponsored clinical trials that many others considered disappointments, without disclosing his relationship to the company. Since 2014, he has received more than $3 million from Roche in consulting fees and for his stake in a company it acquired. Dr. Baselga did not dispute his relationships with at least a dozen companies. In an interview, he said the disclosure lapses were unintentional. He stressed that much of his industry work was publicly known although he declined to provide payment figures from his involvement with some biotech startups. “I acknowledge that there have been inconsistencies, but that’s what it is,” he said. “It’s not that I do not appreciate the importance.” Dr. Baselga’s extensive corporate relationships — and his frequent failure to disclose them — illustrate how permeable the boundaries remain between academic research and industry, and how weakly reporting requirements are enforced by the medical journals and professional societies charged with policing them. A decade ago, a series of scandals involving the secret influence of the pharmaceutical industry on drug research prompted the medical community to beef up its conflict-of-interest disclosure requirements. Ethicists worry that outside entanglements can shape the way studies are designed and medications are prescribed to patients, allowing bias to influence medical practice. Disclosing those connections allows the public, other scientists and doctors to evaluate the research and weigh potential conflicts. “If leaders don’t follow the rules, then we don’t really have rules,” said Dr. Walid Gellad, director of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh. “It says that the rules don’t matter.” The penalties for such ethical lapses are not severe. The cancer research group, the A.A.C.R., warns authors who fill out disclosure forms for its journals that they face a three-year ban on publishing if they are found to have financial relationships that they did not disclose. But the ban is not included in the conflict-of-interest policy posted on its website, and the group said no author had ever been barred. Many journals and professional societies do not check conflicts and simply require authors to correct the record. Officials at the A.A.C.R., the American Society of Clinical Oncology and The New England Journal of Medicine said they were looking into Dr. Baselga’s omissions after inquiries from The New York Times and ProPublica. The Lancet declined to say whether it would look into the matter. Christine Hickey, a spokeswoman for Memorial Sloan Kettering, said that Dr. Baselga had properly informed the hospital of his outside industry work and that it was Dr. Baselga’s responsibility to disclose such relationships to entities like medical journals. The cancer center, she said, “has a rigorous and comprehensive compliance program in place to promote honesty and objectivity in scientific research.” Asked if he planned to correct his disclosures, Dr. Baselga asked reporters what they would recommend. In a statement several days later, he said he would correct his conflict-of-interest reporting for 17 articles, including in The New England Journal of Medicine, The Lancet and the publication he edits, Cancer Discovery. He said that he did not believe disclosure was required for dozens of other articles detailing early stages of research. “I have spent my career caring for cancer patients and bringing new therapies to the clinic with the goal of extending and saving lives,” Dr. Baselga said in the statement. “While I have been inconsistent with disclosures and acknowledge that fact, that is a far cry from compromising my responsibilities as a physician, as a scientist and as a clinical leader.” The corporate imprint on cancer researcher Dr. Baselga, 59, supervises clinical operations at Memorial Sloan Kettering, one of the nation’s top cancer centers, and wields influence over the lives of patients and companies wishing to conduct trials there. He was paid more than $1.5 million in compensation by the cancer center in 2016, according to the hospital’s latest available tax disclosures, but that does not include his consulting or board fees from outside companies. Many top medical researchers have ties to the for-profit health care industry, and some overlap is seen as a good thing — after all, these are the companies charged with developing the drugs, medical devices and diagnostic tests of the future. Dr. Baselga’s relationship to industry is extensive. In addition to sitting on the board of Bristol-Myers Squibb, he is a director of Varian Medical Systems, which sells radiation equipment and for whom Memorial Sloan Kettering is a client. In all, Dr. Baselga has served on the boards of at least six companies since 2013, positions that have required him to assume a fiduciary responsibility to protect the interests of those companies, even as he oversees the cancer center’s medical operations. The hospital and Dr. Baselga said steps had been taken to prevent him from having a say in any business between the cancer center and the companies on whose boards he sits. The chief executive of Memorial Sloan Kettering, Dr. Craig B. Thompson, settled lawsuits several years ago that were filed by the University of Pennsylvania and an affiliated research center. They contended that he hid research conducted while he was at Penn to start a new company, Agios Pharmaceuticals, and did not share the earnings. Dr. Thompson disputed the allegations. He now sits on the board of Merck, which manufactures Keytruda, a blockbuster cancer therapy. Ms. Hickey said the cancer center cannot fulfill its charitable mission without working with industry. “We encourage collaboration and are proud that our work has led to the approval of novel, lifesaving cancer treatments for patients around the world,” she said. Some disclosures are required; others aren’t. After the scandals a decade ago over lack of disclosure, the federal government began requiring drug and device manufacturers to publicly disclose payments to doctors in 2013. From August 2013 through 2017, Dr. Baselga received nearly $3.5 million from nine companies, according to the federal Open Payments database, which compiles disclosures filed by drug and device companies. Dr. Baselga has disclosed in other forums investments and advisory roles in biotech start-ups, but he declined to provide a tally of financial interests in those firms. Companies that have not received approval from the Food and Drug Administration for their products — projects still in the testing phases — do not have to report payments they make to doctors. Serving on boards can be lucrative. In 2017, he received $260,000 in cash and stock awards to sit on Varian’s board of directors, according to the company’s corporate filings. ProPublica and The Times analyzed Dr. Baselga’s publications in medical journals since 2013, the year he joined Memorial Sloan Kettering. He failed to disclose any industry relationships in more than 100, or about 60 percent of the time, a figure that has increased with each passing year. Last year, he did not list any potential conflicts in 87 percent of the articles that he wrote or co-wrote. Dr. Baselga compiled a color-coded list of his articles and offered a different interpretation. Sixty-two of the papers for which he did not disclose any potential conflict represented “conceptual, basic laboratory or translational work,” and did not require one, he said. Questions could be raised about others, he said, but he added that most “had no clinical nor financial implications.” That left the 17 papers he plans to correct. Early-stage research often carries financial weight because it helps companies decide whether to move ahead with a product. In about two-thirds of Dr. Balsega’s articles that lacked details of his industry ties, one or more of his co-authors listed theirs. In 2015, Dr. Baselga published an article in the New England Journal about a Roche-sponsored trial of one of the company’s drugs, Zelboraf. Despite his financial ties to Roche, he declared that he had “nothing to disclose.” Fourteen of his co-authors reported ties to Roche. Dr. Baselga defended the articles, saying that “these are high-quality manuscripts reporting on important clinical trials that led to a better understanding of cancer treatments.” Industry ConnectionsSome of Dr. José Baselga’s known relationships with health care companies. He has failed to disclose any industry ties in dozens of research articles since 2013. The guidelines enacted by most major medical journals and professional societies ask authors and presenters to list recent financial relationships that could pose a conflict. But much of this reporting still relies on the honor system. A study in August in the journal JAMA Oncology found that one-third of authors in a sample of cancer trials did not report all payments from the studies’ sponsors. “We don’t routinely check because we don’t have those kind of resources,” said Dr. Rita F. Redberg, the editor of JAMA Internal Medicine, who has been critical of the influence of industry on medical practice. “We rely on trust and integrity. It’s kind of an assumed part of the professional relationship.” Jennifer Zeis, a spokeswoman for The New England Journal of Medicine, said in an email that it had now asked Dr. Baselga to amend his disclosures. She said the journal planned to overhaul its tracking of industry relationships. The American Association for Cancer Research said it had begun an “extensive review” of the disclosure forms submitted by Dr. Baselga. It said that it had never barred an author from publishing, and that “such an action would be necessary only in cases of egregious, consistent violations of the rules.” Among the most prominent relationships that Dr. Baselga has often failed to disclose is with the Swiss pharmaceutical giant Roche and its United States subsidiary Genentech. In June 2017, at the annual meeting of the American Society of Clinical Oncology in Chicago, Dr. Baselga spoke at a Roche-sponsored investor event about study results that the company had been counting on to persuade oncologists to move patients from Herceptin — which was facing competition from cheaper alternatives — to a combination treatment involving Herceptin and a newer, more expensive drug, Perjeta. The results were so underwhelming that Roche’s stock fell 5 percent on the news. One analyst described the results as a “lead balloon,” and an editorial in The New England Journal called it a “disappointment.” Dr. Baselga, however, told analysts that critiques were “weird” and “strange.” This June, at the same cancer conference, Dr. Baselga struck an upbeat note about the results of a Roche trial of the drug taselisib, saying in a blog post published on the cancer center website that the results were “incredibly exciting” while conceding the side effects from the drug were high. That same day, Roche announced it was scrapping plans to develop the drug. The news was another disappointment involving the class of drugs called PI3K inhibitors, which is a major focus of Dr. Baselga’s current research. In neither case did Dr. Baselga reveal that his ties to Roche and Genentech went beyond serving as a trial investigator. In 2014, Roche acquired Seragon, a cancer research company in which Dr. Baselga had an ownership stake, for $725 million. Dr. Baselga received more than $3 million in 2014 and 2015 for his stake in the company, according to the federal Open Payments database. From 2013 to 2017, Roche also paid Dr. Baselga more than $50,000 in consulting fees, according to the database. These details were not included in the conflict-of-interest statements that are required of all presenters at the American Society of Clinical Oncology conference, although he did disclose ownership interests and consulting relationships with several other companies in the prior two years. ASCO said it would conduct an internal review of Dr. Baselga’s disclosures and would refer the findings to a panel. Dr. Baselga said that he played no role in the Seragon acquisition, and that he had cut ties with Roche since joining the board of a competitor, Bristol-Myers, in March. As for his presentations at the ASCO meetings in the last two years, he said he had also noted shortcomings in the studies. The combination of Perjeta with Herceptin was later approved by the F.D.A. for certain high-risk patients. As for taselisib, Dr. Baselga stands by his belief that the PI3K class of drugs will be an important target for fighting cancer. ReferencesOrnstein, C & Thomas, K. (2018). Top Cancer Researcher Fails to Disclose Corporate Financial Ties in Major Research Journals. [online] Retrieved from: https://www.nytimes.com/2018/09/08/health/jose-baselga-cancer-memorial-sloan-kettering.html
The anti-cancer assertions made by medical cannabis advocates are no longer just based on anecdotal success stories. In the following video, Dr. Christina Sanchez, a molecular biologist at the Complutense University of Madrid, explains how her research supports the claim that cannabis kills cancer. Through several human and animal experimental observations, researchers, such as Dr. Sanchez, have discovered that the application of the popular psychoactive compound found in cannabis, tetrahydrocannabinol (THC), has the potential to induce apoptosis in carcinogenic cells; in other words, THC kills cancer cells. This treatment significantly differs from other cancer treatments, such as chemotheraphy, because THC is able to distinguish between healthy and unhealthy cells, whereas chemotherapeutic agents cannot, resulting in a myriad of undesirable and deleterious side effects. According to Dr. Sanchez, "One of the advantages of cannabinoids, or cannabinoid based medicines, would be that they target a specifically, tumor cells. They don’t have any toxic effect on normal, non-tumoral cells. This is an advantage with respect to standard chemotherapy that target basically everything." Sanchez has further revealed that the cancer-killing effect of THC is potentiated by the presence of cannabidiol (CBD), a very potent antioxidant that protects the brain from stress and oxidative damage. These synergistic effects of both THC and CBD could make for a very safe and effective cancer concoction. Sanchez states, "I cannot understand why in the states cannabis is under schedule I, because it is pretty obvious not only from our work but from work from many other researchers that the plant has very wide therapeutic potential." Cannabinoids and the Endocannabinoid System The human body responds to the cannabinoids found in cannabis because it also makes its own endogenous cannabinoids. These endocannabinoids, which are lipid-based retrograde neurotransmitters that bind to cannabinoid receptors and proteins that are expressed throughout the mammalian central and peripheral nervous system, have been observed to mediate homeostasis via a variety of physiological and cognitive processes. This endocannabinoid system, named after the plant that led to its discovery, is a biological system of the body has many important regulatory functions affecting fertility, pregnancy, reproduction, appetite, pain-sensation, mood, locomotor behavior, exercise-induced euphoria, and memory. The endogenous cannabinoid system is perhaps the most important physiologic system involved in establishing and maintaining human health. Endocannabinoids and their receptors are found throughout the body: in the brain, organs, connective tissues, glands, and immune cells. In each tissue, the cannabinoid system performs different tasks, but the goal is always the same: homeostasis, the maintenance of a stable internal environment despite fluctuations in the external environment. A growing body of evidence continues to validate the miraculous healing capabilities of cannabinoids. Supporting the cultivation of cannabis and continued scientific research will lead to more availability of various cannabis strains specifically designed to have high doses and specific ratios of certain cannabinoids to serve as a safe and effective therapeutic treatment for a variety of disorders and diseases. The use of medical marijuana and cannabis-derived medicines in the treatment of diseases such as cancer, epilepsy, chronic pain, diabetes, multiple sclerosis, insomnia, depression, etc., will continue to grow. Cannabis may yet become one of the most useful natural remedies to some of the most crippling diseases of mankind. ReferencesHunt, Anna. (2018). Molecular Biologist Explains How Cannabis Kills Cancer Cells. [online] Waking Times. Available at: http://www.wakingtimes.com/2017/12/21/molecular-biologist-explains-cannabis-kills-cancer-cells/ [Accessed 6 Jul. 2018].
Physiologically, fasting:
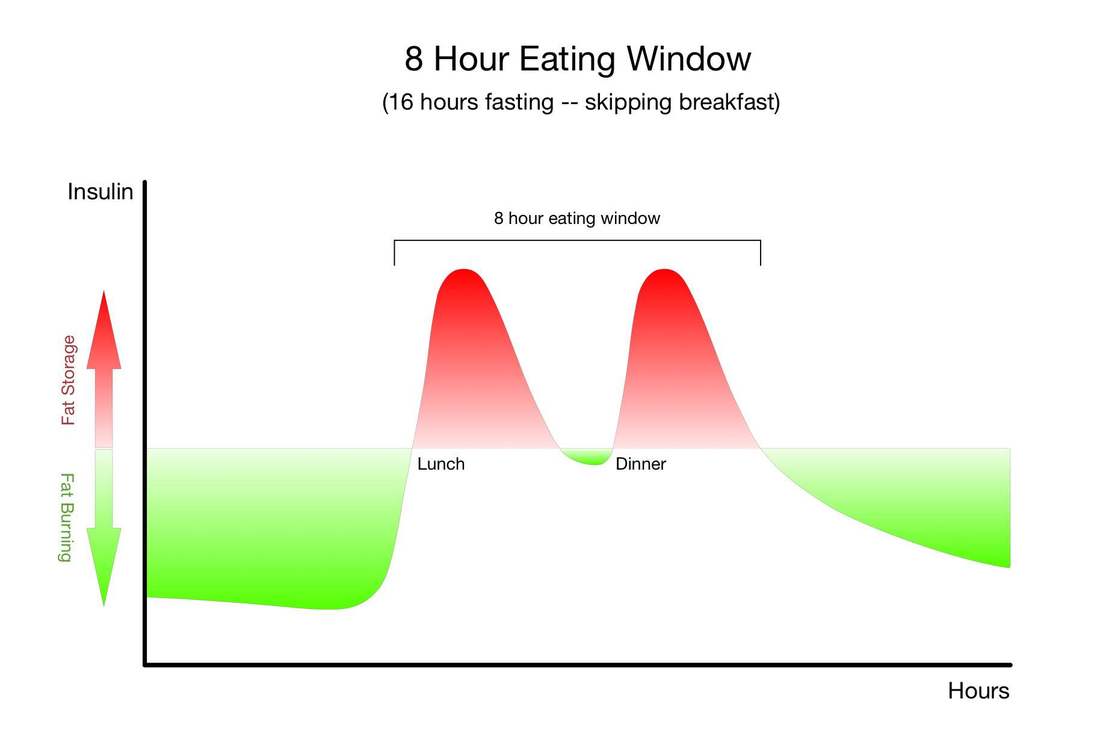
The only other strategy that has so many research-baked benefits for longevity is long-term calorie restriction, which requires a significant long-term reduction in the amount of food you eat so that you are essentially living on the brink of starvation. Compliance with calorie-restricted diets is abysmal. Fortunately, there are many ways to fast, and there is likely a form of fasting out there that you will be able to tolerate and incorporate into your life. It's important for you to remember that fasting can provide nearly identical benefits without the pain, suffering, and compliance challenges of calorie restriction. Instead of regulating how much food you eat, as with long-term calorie restriction, you only need to modify when you eat - and of course wisely choose the foods you do eat.Simply cycling between periods of eating and fasting on a daily, weekly, or monthly schedule has been shown to provide many of the same benefits as long-term calorie restriction. Choosing when to eat and when to fast in this way is known as "intermittent fasting." "Don't eat less - eat less often." |
| Each time you become exposed the warm sunlight, the sun's ultraviolet radiation bombards your skin, which in moderation may optimize your health, or in excess, may lead to sunburns, rapidly aging skin, and potentially, skin cancer. To lower one's risk, many will often turn to sunscreens—sprays, lotions, and even powders—to protect their skin from the worst of the sun's effects. Unfortunately, researchers have collected data showing that you should be very cautious when choosing sunscreen to apply to your skin. |
Skin Cancer on the Rise - No Proof That Sunscreen Prevents Skin Cancer
While the exact cause of melanoma is unknown, researchers have established that risk factors for melanoma include family history, indoor tanning, the number of moles on a person’s skin, fair skin, freckles, blue or green eyes, blonde or red hair and a history of severe sunburns, among others (Centers for Disease Control and Prevention, 2017). People are able to modify only three of these risk factors: indoor tanning, exposure to UV radiation and severe sunburns.
In December 2012, the Food and Drug Administration began to enforce new laws designed to improve sunscreens and consumer protection. The new laws restrict certain bogus label claims, but they allow most sunscreens to advertise “broad spectrum” skin protection. Sunscreen manufacturers are permitted to tell consumers, that with proper use, their products can help reduce the risk of skin cancer. However, the FDA and the International Agency for Research on Cancer have concluded that the available data does not support the assertion that sunscreens alone reduce the rate of skin cancer (Food and Drug Administration, 2011; IARC 2001).
It's Not the Sunscreen, It's the Additives
Researchers have observed that adults who put on sunscreen containing 4% oxybenzone (the US allows up to 6%) in the morning and evening—mimicking what they’d do while on vacation—continued to excrete the chemical in their urine for five days afterwards, suggesting that it was being stored in the body (Gustavsson Gonzalez, Farbrot & Larko, 2002).
Aside from oxybenzone — which is found in 70% of sunscreens — other commonly used chemicals that can enter your bloodstream and can cause toxic side effects, including hormone disruption, include but are not limited to:
Octyl methoxycinnamate (OMC) |
Para-aminobenzoic acid (PABA) |
Octyl salicyclate |
Phenylbenzimidazole |
Octocrylene |
Octisalate |
Dioxybenzone |
Menthyl anthranilate |
Homosalate |
Octinoxate |
Cinoxate |
Parabens |
It is also advised to avoid using personal care products that contain synthetic fragrance, as this term describes any number of harmful chemicals that do not have to be listed individually on the label. Some common "fragrance" chemicals include:
- Parabens: Synthetic preservatives known to interfere with hormone production and release.
- Phthalates: Another synthetic preservative that's carcinogenic and linked to reproductive effects (i.e., decreased sperm counts, early breast development, and birth defects) and liver and kidney damage.
- Synthetic musks: These are linked to hormone disruption and are thought to persist and accumulate in breast milk, body fat, umbilical cord blood, and the environment.
Mineral Sunscreens May Contain Nanoparticles
This nanotechnology has several different effects. Some are concerned that the particles have become so small that they may be absorbed directly into your skin. Although mixed, some studies have found significant negative health effects from the absorption of nanoparticles (European Union Public Health, 2006). While these nanoparticle technologies may make an excellent drug delivery system, it is questionable for use in sunscreen (De Jong & Borm, 2008).
Titanium dioxide is more effective in UVB range and zinc oxide in the UVA range, therefore the combination of these particles assures a broad-band UV protection (Smijs & Pavel, 2011). Zinc oxide is beneficial because it remains stable in heat, but as a nanoparticle, the problems with toxicity probably outweigh the benefits to sun protection. Upon systemic distribution, toxicity of zinc oxide nanoparticles may affect the lungs, liver, kidneys, stomach, pancreas, spleen, heart and brain (Tian et al., 2015). Findings have also demonstrated that aging has a synergistic effect with zinc oxide nanoparticles on systemic inflammation and neurotoxicity, affecting your brain and neurological system. In other words, the older you are, the higher your risk of neurotoxicity from zinc oxide nanoparticle absorption.
Spray-on sunscreens, containing zinc oxide and/or titanium dioxide, pose an additional hazard by releasing these toxic nanoparticles into the air. The FDA has previously expressed concern that inhaling these products may be risky, especially to children, and has warned parents to avoid spray-on sunscreens (Food and Drug Administration, 2006).
While these two minerals are the safest topical sunscreen agents around, inhaling them is a whole different story. When these minerals are inhaled, they have been shown to irritate lung tissues and potentially lead to serious health problems, and the finer the particles, the worse their effects appear to be (Grassian, O’Shaughnessy, Adamcakova-Dodd, Pettibone & Thorne, 2006). The lungs have difficulty clearing small particles, and the particles may pass from the lungs into the bloodstream. Insoluble nanoparticles that penetrate skin or lung tissue can cause extensive organ damage. Some researchers speculate that the toxic effects of nanoparticles relate to their size being in the range of a virus, which may trigger your body's immune response (Buzea, Pacheco Blandino & Robbie, 2007).
The International Agency for Research on Cancer (IARC) has classified titanium dioxide as a "possible carcinogen" when inhaled in high doses. According to IARC:
"Titanium dioxide causes varying degrees of inflammation and associated pulmonary effects including lung epithelial cell injury, cholesterol granulomas and fibrosis. Rodents experience stronger pulmonary effects after exposure to ultrafine titanium dioxide particles compared with fine particles on a mass basis (IARC, 2006).
The use of nanoparticles in cosmetics poses a regulatory challenge because the properties of nanoparticles may vary tremendously, depending on their size, shape, surface area and coatings. A number of manufacturers sell products advertised as containing “non-nano” zinc oxide and titanium dioxide - these claims are generally misleading. While particle sizes vary among manufacturers, nearly all would be considered nanomaterials under a broad definition of the term, including the definition proposed in 2011 by the Food and Drug Administration (Food and Drug Administration, 2011b). According to the available information, these mineral sunscreens must be delivered in nanoparticle form to render a layer that is reasonably transparent on the skin.
According to EWG, even with the existing uncertainties, zinc oxide and titanium dioxide lotions are among the best choices on the American market. Here’s why:
- The shape and size of the particles affect sun protection.
- Nanoparticles in sunscreen may not penetrate the skin.
- It is unlikely that nanoparticles in sunscreen cause skin damage when energized by sunlight.
Currently, all available evidence suggests that zinc oxide and titanium dioxide can be safely used in sunscreen lotions applied to healthy skin and pose a lower hazard than most other approved sunscreen ingredients.
More human studies need to be conducted in regards to the health effects of inhaling of zinc oxide particles, especially at lower levels, such as from brief exposure to sunscreen spray. However, using these spray-on products are clearly an unnecessary risk since safer options are readily available. Your safest bet is to use topical zinc oxide or titanium dioxide that does not contain nanosized particles.
High SPF is "Inherently Misleading"
But for high-SPF sunscreens, theory and reality are two different things. Researchers have observed that people are misled by the claims on high-SPF sunscreen bottles. They are more likely to use high-SPF products improperly and as a result may expose themselves to more harmful ultraviolet radiation than people relying on products with lower SPF values. The FDA has long contended that SPF higher than 50 is “inherently misleading” (Food and Drug Administration, 2007).
Here are some reasons against applying sunscreens with SPF values greater than 50:
- Marginally better sunburn protection.
- Consumers misuse high-SPF products.
Researchers have conducted numerous studies on sunbathers and have observed that high-SPF products spur “profound changes in sun behavior” that may account for the increased melanoma risk found in some studies. The researchers confirmed that European vacationers spent more total time in the sun if they were given an SPF 30 sunscreen instead of an SPF 10 product (Autier et al., 2000). It is assumed the difference would also apply to products with SPF values greater than 50.
Solutions: Sunscreen Should be the Last Resort
Astaxanthin is produced by the microalgae Haematococcus pluvialis when its water supply dries up, forcing it to protect itself from ultraviolet radiation. It is this "radiation shield" mechanism that helps explain how astaxanthin can help protect you from similar radiation.
When you consume this pigment, you are essentially creating your own "internal sunscreen." Researchers have confirmed astaxanthin is a potent UVB absorber that helps reduce DNA damage, reduce inflammation, oxidative stress and free radical damage throughout your body.
Each of these health-promoting effects of astaxanthin improves the ability of your skin to handle sun without burning, while giving your body the best advantage to manufacturing vitamin D. However, it is still advised to seek physical protection from the sun, such as hat and long-sleeved clothing, but consuming more astaxanthin will provide a healthier alternative to using synthetic chemicals to filter UV radiation.
Wear Clothes Shorts, hats, shorts and pants can shield you skin from the sun's UV rays, reducing sun burns by 27% |
Plan Around the Sun Go outdoors in the early morning or late afternoon when the sun is lower in the sky |
Find Shade - Or Make It Find a tree or bring a canopy |
Don't Get Burned Red, sore, blistered skin means you have gotten far too much sun exposure |
Sunglasses Are Essential Polarized sunglasses protect your eyes from the sun's harmful UV rays |
Check the UV Index The UV index provides helpful information to help you plan your outdoor activities without the risk of too much sun exposure |
When Purchasing Sunscreen
References
Autier, P., Doré, J.-F., Reis, A. C., Grivegnée, A., Ollivaud, L., Truchetet, F., … Césarini, J.-P. (2000). Sunscreen use and intentional exposure to ultraviolet A and B radiation: a double blind randomized trial using personal dosimeters. British Journal of Cancer, 83(9), 1243–1248. http://doi.org/10.1054/bjoc.2000.1429
Buzea, C., Pacheco Blandino, I., & Robbie, K. (2007). Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases. Retrieved 3 August 2017, from https://arxiv.org/ftp/arxiv/papers/0801/0801.3280.pdf
Centers for Disease Control and Prevention. (2017). What Are the Risk Factors for Skin Cancer?. Cdc.gov. Retrieved 3 August 2017, from https://www.cdc.gov/cancer/skin/basic_info/risk_factors.htm
Centers for Disease Control and Prevention. (2016). Skin Cancer Trends. (2017). Cdc.gov. Retrieved 3 August 2017, from https://www.cdc.gov/cancer/skin/statistics/trends.htm
De Jong, W. H., & Borm, P. J. (2008). Drug delivery and nanoparticles: Applications and hazards. International Journal of Nanomedicine, 3(2), 133–149.
Environmental Working Group. (2017). EWG's 2017 Guide to Safer Sunscreens. Ewg.org. Retrieved 3 August 2017, from http://www.ewg.org/sunscreen/report/the-trouble-with-sunscreen-chemicals/#.WYKSS4UnHPp
European Union Public Health. (2006). What are potential harmful effects of nanoparticles?. Ec.europa.eu. Retrieved 3 August 2017, from http://ec.europa.eu/health/scientific_committees/opinions_layman/en/nanotechnologies/l-3/6-health-effects-nanoparticles.htm
Food and Drug Administration. (2011). Labeling and Effectiveness Testing: Sunscreen Drug Products for Over-the-Counter Human Use. Regulations.gov. Retrieved 3 August 2017, from https://www.regulations.gov/document?D=FDA-1978-N-0018-0698
Food and Drug Administration. (2011b). FDA Draft, Not for Implementation: Guidance for Industry. Enforcement Policy – OTC Sunscreen Drug Products Marketed Without an Approved Application. Available at www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM259001.pdf
Food and Drug Administration. (2006). PETITION REQUESTING FDA AMEND ITS REGULATIONS FOR PRODUCTS COMPOSED OF ENGINEERED NANOPARTICLES GENERALLY AND SUNSCREEN DRUG PRODUCTS COMPOSED OF ENGINEERED NANOPARTICLES SPECIFICALLY. FDA.gov. Retrieved 3 August 2017, from https://www.fda.gov/ohrms/dockets/dockets/06p0210/06p-0210-cp00001-01-vol1.pdf
Food and Drug Administration. (2007). Sunscreen Drug Products for Over-the-Counter Human Use; Proposed Amendment of Final Monograph; Proposed Rule - FR Doc 07-4131. (2017). Fda.gov. Retrieved 3 August 2017, from https://www.fda.gov/OHRMS/DOCKETS/98fr/07-4131.htm
Grassian, V., O’Shaughnessy, P., Adamcakova-Dodd, A., Pettibone, J., & Thorne, P. (2006). Inhalation Exposure Study of Titanium Dioxide Nanoparticles with a Primary Particle Size of 2 to 5 nm. Environmental Health Perspectives, 115(3), 397-402. http://dx.doi.org/10.1289/ehp.9469
Gustavsson Gonzalez, H., Farbrot, A., & Larko, O. (2002). Percutaneous absorption of benzophenone-3, a common component of topical sunscreens. Clinical And Experimental Dermatology, 27(8), 691-694. http://dx.doi.org/10.1046/j.1365-2230.2002.01095.x
International Agency for Research on Cancer (IARC). (2006). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. IARC.fr. Retrieved 3 August 2017, from https://monographs.iarc.fr/ENG/Monographs/vol93/mono93.pdf
IARC. (2001). IARC Summary Recommendations for Public Health Action. IARC. Retrieved 3 August 2017, from http://www.who.int/uv/resources/recommendations/en/IARCSum.pdf
Mercola, J. (2017). Is Your Sunscreen Doing More Harm Than Good?. Mercola.com. Retrieved 3 August 2017, from http://articles.mercola.com/sites/articles/archive/2017/04/26/hazardous-chemicals-in-sunscreens.aspx
National Cancer Institute. (2017). Melanoma of the Skin - Cancer Stat Facts. Seer.cancer.gov. Retrieved 3 August 2017, from http://seer.cancer.gov/statfacts/html/melan.html
National Toxicology Program (NTP). (2000). Broad-Spectrum Ultraviolet (UV) Radiation and UVA, and UVB, and UVC. U.S. Department of Health and Human Services. Retrieved 3 August 2017, from https://ntp.niehs.nih.gov/ntp/newhomeroc/roc10/uv_no_appendices_508.pdf
NTP Technical Report on the Photococarcinogenesis Study of Retinoic Acid and Retinyl Palmitate [CAS Nos. 302-79-4 (All-Trans-Retinoic Acid) and 79-81-2 (All-Trans-Retinyl Palmitate)] in SKH-1 Mice (Simulated Solar Light And Topical Application Study). NTP TR 568, National Institutes of Health, 2012. Available at ntp.niehs.nih.gov/ntp/htdocs/LT_rpts/TR568_508.pdf
Popov, A., Haag, S., Meinke, M., Lademann, J., Priezzhev, A., & Myllylä, R. (2009). Effect of size of TiO[sub 2] nanoparticles applied onto glass slide and porcine skin on generation of free radicals under ultraviolet irradiation. Journal Of Biomedical Optics, 14(2), 021011. http://dx.doi.org/10.1117/1.3078802
SCCNFP, Opinion Concerning Titanium Dioxide. Opinion: European Commission – The Scientific Committee on Cosmetic Products and Non-Food Products Intended for Consumers. (2000). Retrieved 3 August 2017, from http://ec.europa.eu/health/ph_risk/committees/sccp/documents/out135_en.pdf
Scientific Committee on Consumer Safety. (2012). Opinion on Zinc oxide (nano form). Retrieved 3 August 2017, from http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_103.pdf
Smijs, T. G., & Pavel, S. (2011). Titanium dioxide and zinc oxide nanoparticles in sunscreens: focus on their safety and effectiveness. Nanotechnology, Science and Applications, 4, 95–112. http://doi.org/10.2147/NSA.S19419
Tian, L., Lin, B., Wu, L., Li, K., Liu, H., & Yan, J. et al. (2015). Neurotoxicity induced by zinc oxide nanoparticles: age-related differences and interaction. Scientific Reports, 5(1). http://dx.doi.org/10.1038/srep16117
| Breast cancer is one of the most common forms of cancer. Metastasis, or the development of a secondary malignant growth distance from a primary site of cancer, greatly increases the risk of death from cancer. Researchers have investigated the entry of cancer cells in the blood vessels, a process known as intravasation, and discovered that chemotherapy treatment can also increase this process causing the malignant cells to migrate. In other words, chemotherapy may increase the aggressiveness of cancer by spreading it throughout the body - thereby increasing the disease's fatality. |
Dr. Karagiannis, the lead author of the study, found the number of doorways was increased in 20 patients receiving two common chemotherapy drugs. It should be noted such a small sample size constitutes a major limitation of this study, the results and implications of this research are very significant.
Solutions
- Lifestyle risk factors, such as tobacco smoking, physical activity, and alcohol use.
- Dietary risk factors, such as a high salt intake or a low intake of fruits and vegetables.
- Metabolic risk factors, such having high blood pressure or blood cholesterol and being overweight or obese.
In regards to dietary interventions, numerous studies have substantiated the chemopreventative effect of increasing cruciferous vegetable intake against cancer. As a chemoprevention agent, sulforaphane, the active molecule in cruciferous vegetables, possesses many advantages, such as high bioavailability and low toxicity (Li et al., 2010).
References
Li, Y., Zhang, T., Korkaya, H., Liu, S., Lee, H., & Newman, B. et al. (2010). Sulforaphane, a Dietary Component of Broccoli/Broccoli Sprouts, Inhibits Breast Cancer Stem Cells. Clinical Cancer Research, 16(9), 2580-2590. http://dx.doi.org/10.1158/1078-0432.ccr-09-2937
Archives
April 2024
March 2024
February 2024
January 2024
December 2023
September 2023
August 2023
June 2023
October 2022
July 2022
February 2022
January 2022
December 2021
November 2021
October 2021
August 2021
July 2021
June 2021
March 2021
February 2021
January 2021
December 2020
November 2020
September 2020
August 2020
March 2020
December 2019
November 2019
October 2019
September 2019
August 2019
July 2019
May 2019
April 2019
March 2019
February 2019
January 2019
December 2018
November 2018
October 2018
September 2018
August 2018
July 2018
June 2018
April 2018
March 2018
February 2018
January 2018
December 2017
November 2017
October 2017
September 2017
August 2017
July 2017
June 2017
May 2017
April 2017
March 2017
February 2017
January 2017
Categories
All
5G
Adductors
Anxiety
Autism
Ayurveda
Body
Breathing
Cancer
Cannabis
Carbohydrates
Cardiovascular Disease
Children
Chronic Disease
Cognition
Consciousness
Coronavirus
Covid
Cryotherapy
Depression
Deuterium
Diet
Dietary Guidelines
EMFs
Emotions
Endocrine Disruptors
Environment
Exercise
Farming
Fasting
Fats
Fluoride
Food
Food Like Product
Food-like Product
Forest
Gardening
Genetics
Glyphosate
GMOs
Hamstrings
Healing
Health
Herbalism
Hormones
HRV
Immunity
Infertility
Laughter
Lockdowns
Loneliness
Longevity
Masks
Meditation
Metabolism
Microbiome
Microwave Radiation
Mind
Mortality
Musculoskeletal System
Nature
Neuroplasticity
Nutrition
Omega 3
Omega-3
Organic
Pelvis/Thigh
Pesticides
Physical Therapy
Placebo
Pollution
Positive
Pregnancy
Prevention
Processed Foods
Psi
Quadriceps
Research
Retirement
Salt
Sleep
Spine/Thorax
Spirit
Stress
Sugar
Technology
Touch Screens
Toxicity
Vaccines
Well Being
Well-Being




 RSS Feed
RSS Feed

